LMCB Group Leader, UCL Professor of Developmental Biophysics
MRC Senior Fellow
Deputy Director of UCL IPLS
y.mao@ucl.ac.uk
+44 (0)20 7679 7267
LMCB Room 2.12
UCL Profile | Europe PMC | PubMed | Media | CV & awards | Funding | Resources | Alumni | Lab website
Lister Prize Fellowship Presentation
Twitter @YanlanMao
Research synopsis
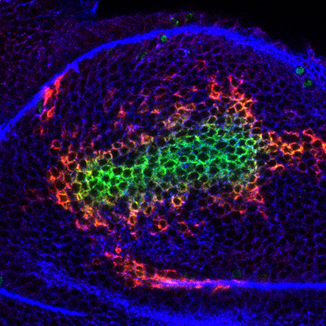
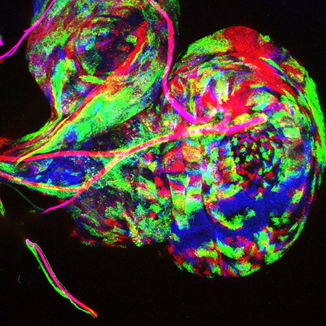
In the Mao lab we are interested in understanding how tissues achieve their correct size, shape and complex three-dimensional architecture, both during normal development, and during regenerative growth.
The genetic and biochemical control of tissue growth and regeneration has been extensively studied over the last century, but it is still unclear how the physical and mechanical properties of cells and tissues contribute to how organs are formed and sculpted. What is clear is that in order to change the three-dimensional architecture of any structure, there must be forces, external and/or internal, acting on the system.
We use an interdisciplinary approach, combining Drosophila genetics, live imaging, automated image analysis, experimental biophysics, engineering and computational modeling, to understand the importance of mechanical forces in controlling tissue growth and regeneration and how these forces in turn influence gene expression and signaling pathways.
Selected publications
About the lab
Funders
Research themes
Technology
People
Ricardo Barrientos (Research Associate)
Alejandra Guzman-Herrera (PhD student)
Collaborators
Jemima Burden (LMCB, UK)
Eileen Gentleman (KCL, UK)
Chris Tape (UCL, UK)
Luigi Aloia (Astra Zeneca, UK)
Julien Vermot (Imperial College London, UK)
Jose Munoz (UPC Barcelona, Spain)
Guillaume Charras (LCN, UCL, UK)
Ewa Paluch (Cambridge University, UK)
Buzz Baum (MRC LMB, UK)
Luis Escudero (Seville University, Spain)
 Close
Close


