Research synopsis
The Saiardi lab is interested in the intersection between signalling and basic metabolism. Specifically, we study the role played by inositol pyrophosphates in maintaining cell homeostasis.
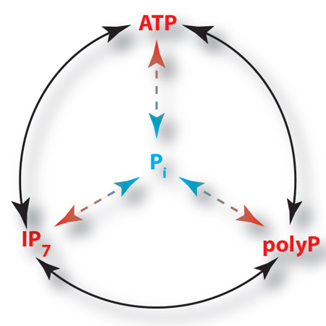
Inositol pyrophosphates are present in all eukaryotic cells and belong to the inositol phosphate family of soluble signaling molecules. Both inositol phosphates and inositol pyrophosphates are obtained by the combinatorial attachment of phosphate groups to inositol. However, the inositol pyrophosphates are distinguished by their one or more high-energy phosphoanhydride bonds (pyro-moieties). The energy stored in these bonds provides the inositol pyrophosphates with the potential to take part in new and not yet fully understood molecular actions.
The structure of these molecules and their conservation in eukaryotic biology suggests a fundamental signalling role, acting through a unique mechanism. Alteration of inositol pyrophosphate levels has been associated with a wide collection of phenotypes as well as important human pathology such as obesity, diabetes and cancer. These pleiotropic effects suggest an ability to control a central cellular function.
Our finding that inositol pyrophosphates regulate intracellular levels of ATP places these molecules at the interface between signalling and basic metabolism. Research in our lab focuses on understanding how inositol pyrophosphates regulate basic metabolism.

Selected publications
Funders
Medical Research Council
Research themes
Technology
People
Filipy Borghi (Research Associate)
Xue (Bessie) Su (Research Associate)
 Close
Close


