Research synopsis
Our research aims to unravel one of the greatest mysteries in biology and medicine – how different cell types interact to generate functional organs during development.
Our focus is on epithelial tissues, the building blocks of most organs. These tissues perform critical functions such as selective exchange of gas, nutrients, and waste products, and provide structural support. They are masterfully shaped to suit each organ's unique needs, from stratified layers for protection, like the skin, to tubular networks for increased surface area, such as in the intestine.
Generating the intricate three-dimensional epithelial structures that support our organs requires cells to coordinate their movement and changes in their shape. Our research aims to uncover the mechanisms and molecular pathways behind this coordination. To study this, we use a multidisciplinary approach that combines genetic, molecular and optogenetic methods in vivo, mammalian cell cultures, mathematical modelling, and state-of-the-art imaging. Our favourite model system is the genetically amenable eye of Drosophila melanogaster, which allows us to perturb gene expression and protein function in individual cell types while studying the consequences in a developing living epithelium.
By understanding how cells work together to induce three-dimensional tissue organisation during organ development, we hope to gain insights into how abnormal epithelial tissues architecture might arise in development and homeostasis, causing diseases such as cancer, for example. The goal of our research is to contribute to our understanding of the pathways that promote epithelial tissue development and to help advancing our ability to diagnose, treat, and prevent epithelial diseases.
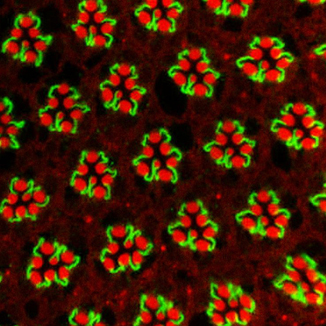
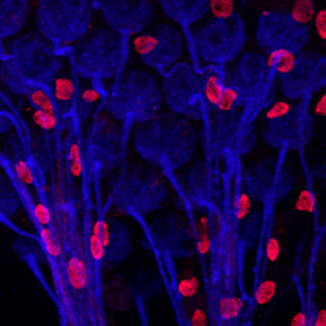
Selected publications
Funders
Research themes
Technology
People
Rhian Walther (Investigator scientist, MRC)
Chantal Roubinet (Postdoctoral researcher, CRUK)
Courtney Lancaster (PhD student, Optical Biology, Wellcome)
Gautum Vasnani (PhD student)
Usman Mukhtar (MSci Cell Biology)
Deborah Matinuzzi (MSci Cell Biology)
 Close
Close


