To ensure regulatory compliance, it is important to engage with regulators in advance to identify and address potential hurdles to translation.
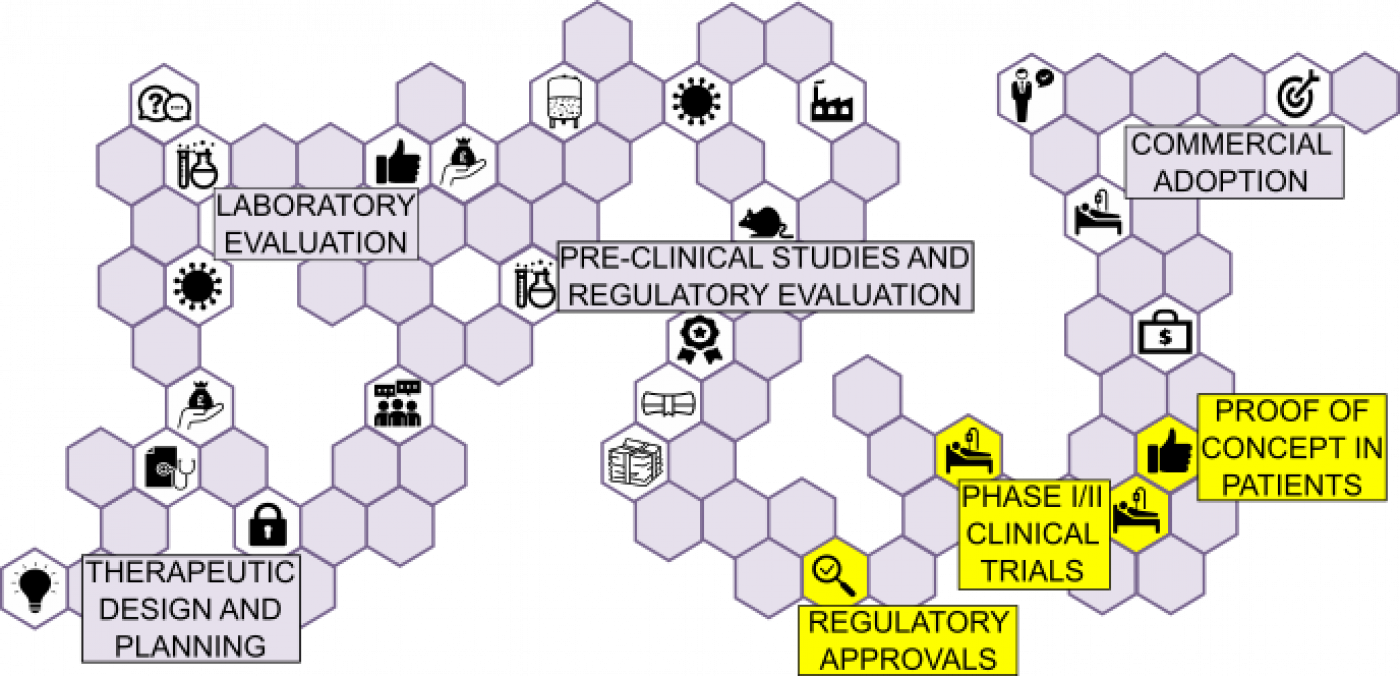
 REGULATORY APPROVALS:
REGULATORY APPROVALS:
It is crucial to achieve regulatory and ethical compliance at the stage of early phase clinical studies.
Considerations include:
- Assign responsibility for generating regulatory documentation and submissions
- Engaging with Regulators in advance to identify and address potential hurdles to translation
- Ensuring rigorous compliance of UCL researchers and contractors to required international standards
- Consideration of the precedent for trial design/adaptive design approaches
- Consider the inclusion of PPI as part of trial design and development.
 PHASE I/II CLINICAL TRIAL:
PHASE I/II CLINICAL TRIAL:
While Phase I clinical trials are for safety, Phase I/II clinical trials can encompass a secondary objective of assessing efficacy, if administered to a patients’ population and can be compared to standard of care practice.
Some important considerations include:
- Drug developers are required to conduct first in human clinical trials to establish the safety of a new therapeutic
- Single/dose escalation study to establish efficacy and expression
- Trial must be compliant with Good Clinical Practice (GCP).
PHASE II CLINICAL TRIAL
Phase II clinical trials objective is to assess safety and efficacy of therapeutic in extended target patient population:
- Participant follow-up (10 years)
- Protocol based upon anticipated real world dosing regimen
- Trials must be compliant with GCP
UCL Support:
There are several clinical trial units that the UCL Translational Research Group can liaise with, including:
- Cancer Research UK & UCL Cancer Trials Centre
- Comprehensive Clinical Trials Unit at UCL
- Medical Research Council Clinical Trials Unit at UCL
- PRIMENT Clinical Trials Unit
The TRG work with the Joint Research Office to handle clinical trial related work.
 Close
Close

