Education Centre

TINs Roadmaps
We have developed roadmaps as tools to map out the development pathway for each therapeutic modality. Within the roadmaps, you are signposted towards the specific internal UCL and external resource that can provide support at each stage of the translational process.
Translational Training - ACCELERATE
Developed by the Academic Careers Office, ACCELERATE is a portfolio of activities aimed at upskilling the UCL research community in translational research through technical training, skills development, and encouraging innovative thinking. Here, also find other relevant training programmes across the university and educational resources beyond UCL.

Informational Resources for Translational Research
Visit the Translational Research Office (TRO) website to view reports, papers, blog articles and other educational resources useful for those wishing to learn more about translational research and the extensive infrastructure in place at UCL to accelerate translation.

TIN Webinar Recordings
We have made our online Therapeutic Innovation Networks events available to view as recorded webinars here.
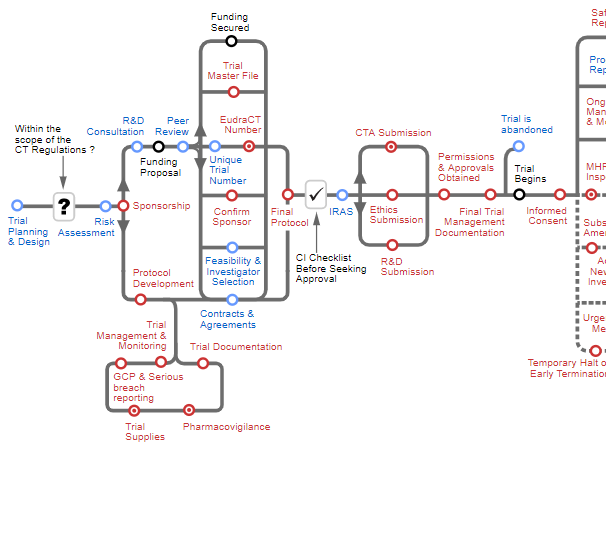
NIHR Clinical Trials Toolkit
Developed by the National Institute for Health Research (NIHR), the Clinical Trials Toolkit provides practical advice to researchers in designing and conducting publicly funded clinical trials in the UK. Through the use of an interactive routemap, this site provides information on best practice and outlines the current legal and practical requirements for conducting clinical trials. [*Disclaimer]

Oxford Global Guidance Devices Regulation Decision Support Tool
An excellent digital tool from Oxford Global aimed at people working in the area of medical devices, helping researchers to navigate device and IVD regulations. [*Disclaimer]

Vigene Viral Delivery Guide - Choose the right viral vector for your research
Vigene designed this guide to help researchers decide which of the three most popular recombinant viruses, AAV, adenovirus, or lentivirus, will work best for their specific applications. [*Disclaimer]

MDC Connects: A Guide to Drug Discovery
In the Spring of 2020, Medicines Discovery Catapult (MDC) ran a series of 9 weekly webinars, MDC Connects, each comprising 2 or 3 different talks. This guide contains a write up summary of each presentation from the webinars, creating a great resource of modern drug discovery knowledge especially useful for the Small Molecules TIN. [*Disclaimer]

MDC Connects: A Guide to Complex Medicines
Another useful guide from the Medicines Discovery Catapult. This time on Complex Medicines (Biologics, ADCs etc.), and is based on the output from their 2021 series of webinars focussed on complex medicines. [*Disclaimer]
In addition to the guide, the "Proposed Definition of ‘Complex Medicine" is a good resource especially for those in the Biologics TIN and can be found here: https://md.catapult.org.uk/resources/proposed-definition-of-complex-medicine/

Report/guide from LifeArc and Pinsent Masons on 'Repurposing drugs: the opportunities and Challenges',
LifeArc and Pinsent Masons have developed a guide to 'Repurposing medicines:the opportunity and the challenges'. The guide includes lessons aimed at supporting charities and researchers that are researching new uses for existing medicines. [*Disclaimer]

Lead optimization: Delivering molecules fit for preclinical development
A careful lead optimization program is critical if a molecule is to progress successfully through preclinical studies and into clinical development. The initial leads that emerge from a hit-to-lead program, while they may show promising early signs of efficacy, may have features or properties that make them less likely to make a successful medicine.
Read more in the FREE e-book published by Fierce Biotech. [*Disclaimer]

The Medicines Repurposing Programme for England by NHS England
The national multi-agency Medicines Repurposing Programme identifies and progresses opportunities to use existing medicines in new ways, outside the current marketing authorisation or licence. This video explains which medicines are eligible, the support on offer, and our first successful licence variation for anastrozole. We are always searching for projects that use medicines in new ways to benefit patients. Source of the video: NHS England [*Disclaimer]
 Close
Close

