Clinical research
Dr Simona Balestrini
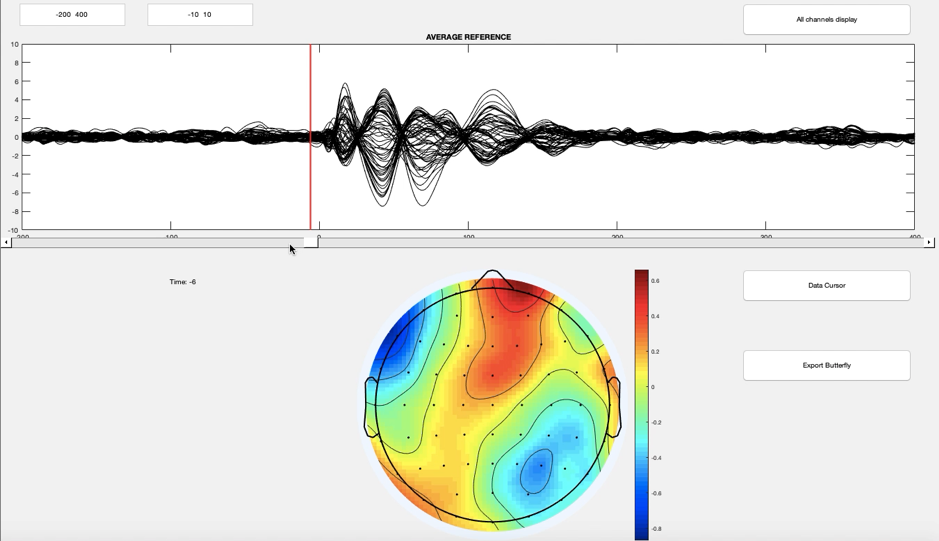
Our lab focuses on application of Transcranial Magnetic Stimulation (TMS) combined with EEG in epilepsy to investigate pathophysiological mechanisms. We are particularly interested in genetic epilepsies, and we use TMS-EEG as a novel translational approach to understand epileptogenesis processes and to establish the neurophysiological profile in individuals with genetic epilepsies, guiding and monitoring individualised treatment options.
Professor Beate Diehl
Professor John Duncan
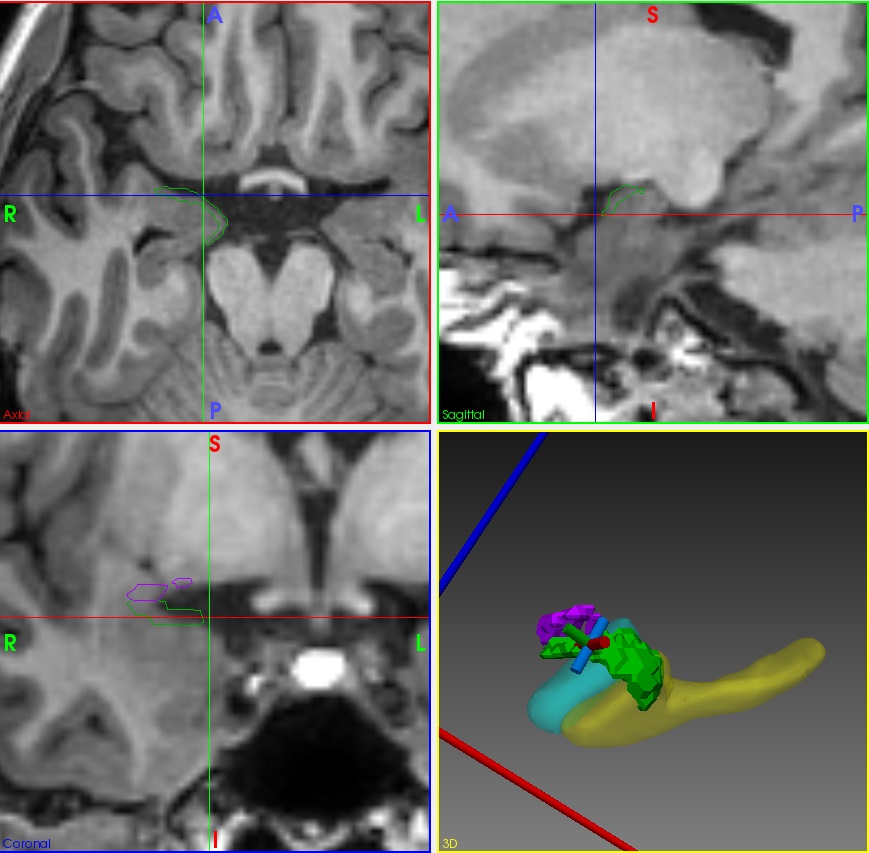
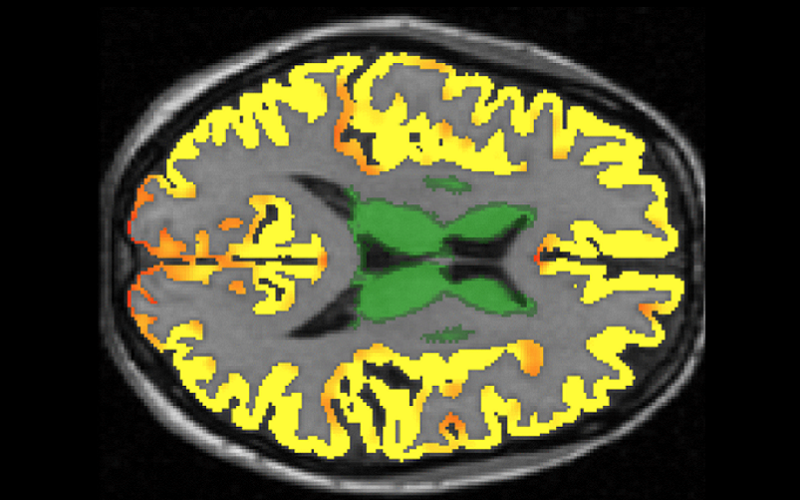
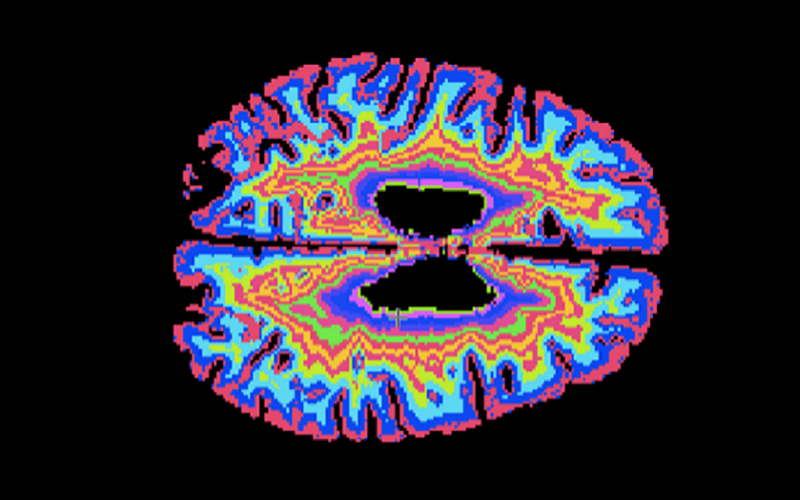
We are pioneering image-guided epilepsy surgery, funded by the Wellcome Trust. We integrate normal and abnormal brain structures in a common 3D space, with the EpiNAv platform that we created with colleagues at KCL. This enables display of eloquent cortex visualized with fMRI, critical tracts demonstrated with high-resolution tractography, vasculature and the parts of the brain that give rise to epileptic seizures. These data are used with intraoperative MRI-based navigation to optimize the chances of seizure freedom and to minimize the risk of complications of neurosurgery.
Professor Matthias Koepp
Professor Louis Lemieux
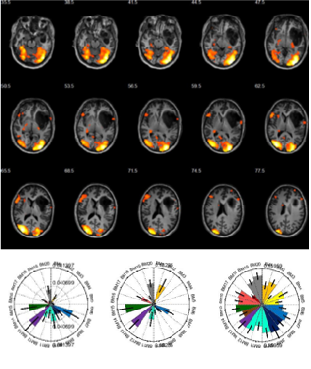
We are working on better understanding the nature of the generators of pathological EEG and functional MRI signals in humans affected by epilepsy, and study the associated brain networks. For this purpose, we develop multi-modal imaging techniques, fused multi-modal data analysis and modelling methods.
Dr Fergus Rugg-Gunn
Professor Ley Sander
Dr Meneka Sidhu
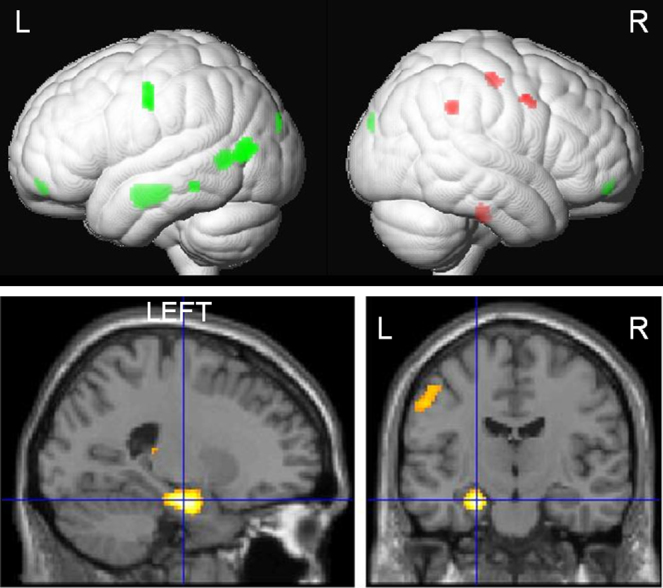
Our work is based on investigating the functional and structural correlates of cognitive processes in people with epilepsy. We use functional magnetic resonance imaging to study language and memory networks involved in these processes with the translational application of improving the precision of epilepsy surgery. These methods have been developed to be used as tools to predict cognitive outcomes after epilepsy surgery. Improving the precision of tasks and the combination of functional and structural data allows for more accurate localisation of eloquent functions, mitigating the risks associated with epilepsy surgery.

Professor Sanjay Sisodiya
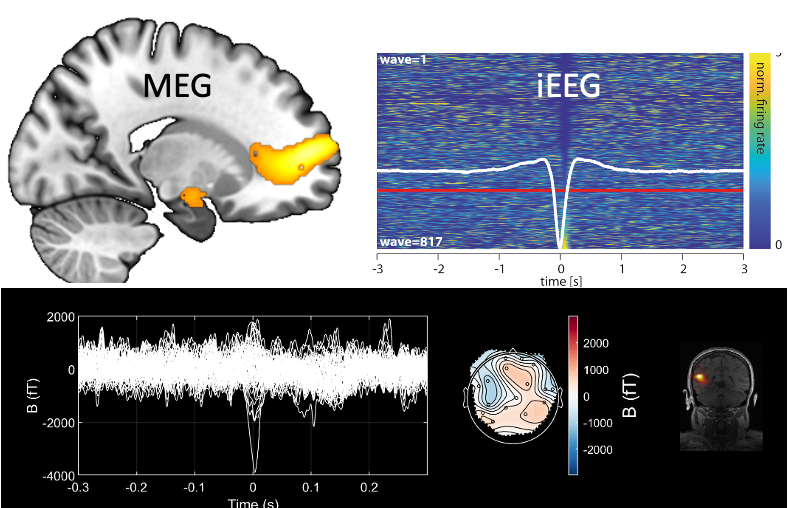
Dr Umesh Vivekananda
Our group focusses on the computational neurophysiology of epilepsy at multiple levels from cellular (animal models/single neuronal recording in humans), to brain region (intracranial EEG) to whole brain (magnetoencephalography in particular OP-MEG). Our aim is to better understand the electrical basis for seizure and epileptiform activity, and its impact on cognition and mood.
 Close
Close

