Study uncovers crucial insights in Ultra-Rare Genetic Syndrome Tied to Potassium Channel Dysfunction
18 March 2024
In an international study co-led by Dr Efthymiou and Professor Houlden at UCL Queen Square Institute of Neurology in collaboration with Rutgers University, novel genetic insights into ABCC9-related intellectual disability and myopathy syndrome (AIMS) have been unveiled.

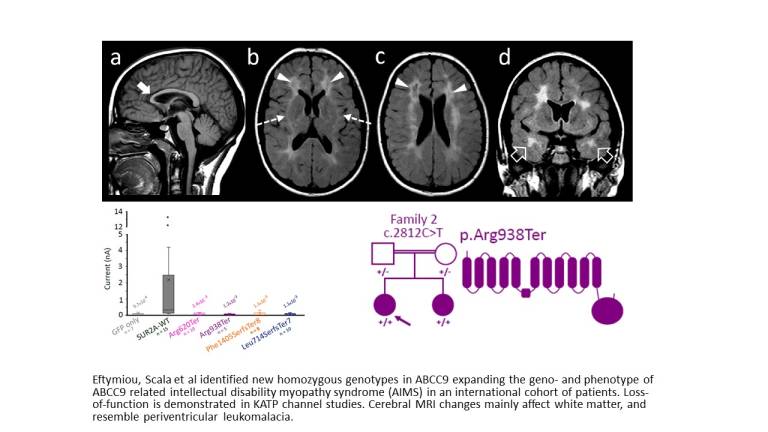
This study published in Brain has significantly expanded our understanding of AIMS by identifying new genetic variants and delineating the disease’s phenotype associated with potassium channel dysfunction.
Utilizing CRISPR/Cas9 genome editing in animal models, the research team have provided a deeper look into the diverse and severe impacts of AIMS mutations, highlighting the potential for variability in patient experiences. Notably, the study revealed critical neurological impairments and brain abnormalities in new patient cohorts, paralleled by zebrafish models exhibiting similar neurological disruptions and increased seizure susceptibility.
These findings mark a substantial advance in the understanding of AIMS, demonstrating the value of integrating patient data with animal model research to explore the full scope of the disease’s impact. By uncovering previously unrecognized aspects of AIMS, including potential cardiac issues, brain white matter involvement and disrupted neural balance, this research not only deepens our comprehension of the syndrome but also opens avenues for future therapeutic strategies.
Dr Conor McClenaghan, collaborator on this study from Rutgers University recently visited to give a seminar on ATP-sensitive potassium channels: From genetic mutations to multi-system disease.
Dr McClenaghan said: “This work has been an excellent example of productive collaboration, enabled by the insights from Stephanie, Marcello Scala, and Henry, and involving vital contributions from colleagues all over the world. This key finding confirms our previous association of loss-of-function variants in ABCC9 with AIMS and promises to lead to greater understanding of KATP channel-associated pathologies, and mechanisms of neurological and myopathic disease, in general. Hopefully, with improved knowledge of how pathology arises in AIMS, and the continued co-operation from our wonderful colleagues, we can move closer to identifying potential treatments for this orphan disease.”
Dr Efthymiou said: “This piece of research on KATP-channel associated AIMS disease was an amazing collaboration between professionals from around the globe pooling their clinical expertise, sharing genetic data, and using the best technologies to model a rare disease. By working together with Conor, Marcello, Marie and the clinical collaborators we were able to uncover novel insights. We are now planning how to take some of these findings from lab bench to bedside, by developing effective treatments.”
Links
- Efthymiou et al. Novel loss-of-function variants expand ABCC9-related intellectual disability and myopathy syndrome, Brain, 2024;, awae010, https://doi.org/10.1093/brain/awae010
- Dr Stephanie Efthymiou's Profile
- Professor Henry Houlden's Profile
Main image: Dr Conor McClenaghan, Dr Stephanie Efthymiou, Professor Henry Houlden
 Close
Close

