 | ______________________________________Dr Rickie Patani is a Clinical Neurologistand Stem Cell Biologist______________________________________Follow us on Twitter: @PataniLab |
Our Research
Neurological disorders include a range of progressive and largely untreatable conditions that are characterized by cellular degeneration in a region- and/or subtype-selective fashion. There is a great experimental need for renewable sources of clinically relevant and regionally defined subclasses of neurons and glia to understand the precise underlying molecular pathogenic events responsible for causing neurodegeneration.
Against this background, our lab focuses on two main themes:
1) Disease Modelling
In order to closely approximate human pathophysiology we integrate the human induced pluripotent stem cell (iPSC) technology together with developmentally rationalised directed differentiation strategies to generate clinically relevant and regionally defined populations of neurons and glia. This serves as an ideal experimental platform to interrogate early pathogenic events in a range of neurodegenerative disorders. The overarching focus of our laboratory is to uncover the precise roles of deregulated RNA metabolism and cellular autonomy in the pathogenesis of motor neuron disease (MND).
2) Developmental Neurobiology
A pre-requisite to harnessing the promise of the iPSC technology in Regenerative Medicine is a comprehensive understanding of cell type specific developmental lineage restriction programmes. We utilise ontogeny-recapitulating differentiation strategies to elucidate key events in neural lineage restriction, subsequent regional fate specification and functional maturation of diverse neuronal and glial subtypes.
_____________________________________________________________________
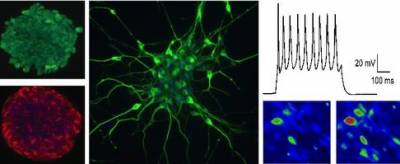
The panel above shows evidence of neural conversion from human pluripotent stem cells. Enriched populations of neural precursors (left column) are subsequently terminally differentiated into neurons (middle image), in which functional maturity is confirmed both electrophysiologically and by calcium imaging methods (right column).
_____________________________________________________________________
Postdoctoral Research Associates
Dr Martina Hallegger (2014-present): Martina is investigating how deregulated RNA metabolism contributes to the earliest events in the molecular pathogenesis of MND. She studies defective pre-mRNA processing by applying high-throughput methods like iCLIP (UV cross linking with immunoprecipitation), RNAseq and proteomics. Her work is funded by the Wellcome Trust and she is co-mentored by Prof. Ule (UCL).
Dr Abi Li (2015-present): Abi is an Honorary Lab Manager and Senior Research Technician. Abi's role involves helping to organise and ensure the smooth running of the laboratory. Abi is a dedicated teacher and facilitator of new students and visitors, specifically with iPSC work. She also conducts research and is currently investigating the pathobiology of paroxysmal kinesigenic dystonia (PKD) using iPSCs.
Dr Zhi Yao (2015-present): Zhi uses various live cell imaging assays together with genome editing methods to comprehensively characterize the phenotype of MND patient specific iPSC-derived spinal motor neurons and glia. She is funded by a Takeda-UCL partnership and co-mentored by Dr Sonia Gandhi (UCL).
Dr Giulia Tyzack (2015-present): Giulia studies neuron-glia interaction in MND patient specific iPSCs. Her work is funded by the Wellcome Trust and she is co-mentored by Dr Andras Lakatos (Cambridge University).
Dr Philip Smethurst (2015-present): Phil is investigating the toxicity and spread of TDP43 protein in iPSC-derived spinal motor neurons. Phil also actively drives public engagement and is partly funded through the Wellcome Trust. He is co-mentored by Dr Katie Sidle.
PhD Students
Miss Claire Hall (2013-present): Claire uses a combination of human iPSC models and functional genomic technologies to understand how different MND-causing mutations perturb RNA metabolism. Claire is funded by UCL Grand Challenges and is co-supervised by Prof. Jernej Ule and Prof. Nicholas Wood.
Dr Rubika Balendra (2013-present): Rubika integrates the human iPSC in-vitro system with a Drosophila in-vivo model to investigate the molecular pathogenic events in MND–causing mutations in the C9orf72 gene. She is co-supervised by Dr Adrian Isaacs and Prof. Linda Partridge and is funded by a Wellcome Trust Research Training Fellowship and the UCL Leonard Wolfson Experimental Neurology Centre.
Dr Sarah Wiethoff (2014-present): Sarah is studying differentiation potential of human iPSCs into cerebellar derivatives and investigates their utility for disease modelling in the spinocerebellar ataxias (SCAs). Sarah's work is funded by Brain Research Trust. She is co-supervised by Prof. Henry Houlden and Prof. John Hardy.
Mr Daniel Cotfas (2015-present): Dan studies beta-propeller protein associated neurodegeneration (BPAN) to ultimately gain insight into highly prevalent forms of neurodegeneration including Parkinson’s and Alzheimer’s diseases. He is co-supervised by Prof. Tom Warner and Dr Abi Li.
Mr Luke Hill (2015-present): Luke studies hereditary spastic paraparesis (HSP) using patient specific human iPSCs. He is co-supervised by Prof. Tom Warner and Dr Abi Li.
Dr Helen Devine (2015-present). Helen is a clinical PhD student investigating the pathogenesis of Spinal and Bulbar Muscular Atrophy (SBMA). She is co-supervised by Prof Linda Greensmith and Prof Michael Hanna.
Research Technician
Dr Alexandra Zirra (2014-present): Alexandra is a medical doctor embarking on a PhD in October 2015. During her time in our lab, she has worked mainly on developmental aspects of neural conversion from iPSCs together with Sarah Wiethoff. In parallel, she works on various functional genomics projects closely supervised by Prof. Ule.
 Close
Close

