Working with Genetically Modified Material (GMM)
All work activities must be assessed. Additional review is required for all work with GMM, even if the agent was prepared elsewhere and the work does not involve further modification.
On this page
> Before starting work
> Guidance on GMM risk assessments
> Guidance on Class 2 and Class 3
> Guidance on Living GMO risk assessments
> Guidance on Lentiviral vectors
> Containment and storage
> Summary of requirements for approval
Before starting work
A risk assessment is required prior to the use of GMOs. The term 'contained use' is any activity in which GMOs are cultured, stored, transported, destroyed, disposed of or used in any other way and for which physical, chemical or biological barriers are used to limit contact with humans or the environment to ensure a high degree of safety.
UCL typically works under the Genetically Modified Organisms (Contained Use) Regulations. However, if GMOs are being released into the environment the Genetically Modified Organisms (Deliberate Release) Regulations should be followed. Email biochem@ucl.ac.uk for more information.
- Work must not start until your assessment has been reviewed and all appropriate authorisations obtained (including any external approvals from HSE) - you will receive an email notification from riskNET indicating when the assessment has been approved.
- Check that the area where your work is to be carried out is already notified to HSE. If not, email biochem@ucl.ac.uk so that the HSE can be notified. Remember to consider work carried out in laboratories and Biological Services Units.
- Carefully consider the scope of your assessment. Where there are a number of activities carried out in contained use that are integrated into a larger programme a 'connected programme of work ' should be considered. This can avoid over-notification of work.
- Check to see if there is already a risk assessment in the system similar to the work proposed.
- If a similar risk assessment exists, the risk assessment can be copied and amended as necessary to cover the proposed work.
Copying an existing risk assessment
- If there is an authorised risk assessment in place that is similar to the work proposed, the risk assessment can be 'copied' creating a new risk assessment with a new reference number which you can amend to reflect the new work.
- If a similar risk assessment does not exist then a new one will need to be created on the relevant template e.g. Class 1, Class 2, Class 3, GM animals, GM plants. The class of the GMM will be determined by the controls required for the activity involving GMM.
- Guidance on completing the GM risk assessments can be found in the SACGM Compendium of guidance. There is also specific guidance on risk assessments in the drop-downs: Guidance on GMM, Class 2 and 3, GM Animal risk assessments. Please remember that others (including the HSE) reviewing the assessment may not be familiar with the subject, and may need more information on the nature of the hazards. The following video is a guide on how to create a new risk assessment. Please select the specialist rsk asssesment form e.g. Class1, 2 or 3. The following video is a guides you through how to create a new risk assessment.
MediaCentral Widget Placeholderhttps://mediacentral.ucl.ac.uk/Player/GB1He3JJ riskNET: Risk Assessment of Biological agents and plants provides guidance on completing and approving biological agent and plant risk assessments, covering genetically modified and wild type work activities.- If you are not sure which assessment forms to use or need help with completing your risk assessment, speak to your local Genetic Modification Safety Officer in the first instance. You can also contact the University BioChem mailbox for further help and advice if required.
- Once the risk assessment is complete select 2 authorisers from your local department. Typically this will be the PI who is in control of the work and the local GMSO. If the Assessor is the PI then a peer with experience and knowledge of the work can review and approve the risk assessment for the technical content. It is the department's responsibility to approve the risk assessment.
- Submit the Risk Assessment for authorisation.
- For Class 2, Class 3, GM animal and GM plant risk assessments there is an additional review completed by the Genetic Modification and Biological Safety Sub Committee (GMBSSC). This is to ensure all the risk assessments have sufficient details of the hazards and controls for the proposed GMO work.
- Once the GMBSSC are satisfied that the content is sufficient all new class 2 and Class 3 risk assessments will be notified to the HSE see notifications for class 2 and class 3 GMM for further information
- When the risk assessment has been approved by the Department, GMBSSC and in some cases the HSE work can start.
- Guidance for GMM risk assessments
All GMM risk assessments should provide an overview of the components of the GMO construct and the activities proposed including the following:
Factors to consider Information required Recipient organism(s) Give the name of the organism(s) to be used including strain, the name of the wild‐type organism it is derived from, provide ACDP HG, degree of disablement/attenuation. Cite evidence. Vector(s) List names of vectors and indicate any disabling mutations including the extent to which they can be mobilized (if relevant). Cite evidence Inserted genetic material List names of genes/genetic material to be inserted. Describe the function of the inserted material (if unknown, consider likely effects by comparison with homologues if available) and the source of the genetic material e.g., sourced from a wild‐type organism, generated artificially or altered in anyway before insertion. If you are looking at groups/families of genes, list all to be used with a description of the function(s) of the gene family. Human health and the environment
Factors to consider Information to be provided The most hazardous GMM to be constructed Consider hazard impact to human health and the environment. Consider wild types, mutations, likelihood of reversion, over expression, toxic, allergenic, oncogenic effects, tropism, survival, and impact to the environment. Risks to human health What is the result of an exposure to human health. Survival, severity of response, likelihood of exposure and any uncertainty Risk to environment Are the organisms covered by additional legislation, infection and colonisation. Could the GMM be an adventitious agent. For Class 2 and 3 GM risk assessments the above information will be required plus more comprehensive information as described in the table below:
Health hazards
Hazards of: Information required The recipient organism ACDP HG of the wild type, mode of transmission, disease symptoms, tissue tropism. Availability of vaccines or therapeutics treatments Alterations Pathogenicity, adhesion / penetration factors or surface components (receptors, envelope protein, capsids) providing resistance to immune system or therapeutic drugs. Inserts and or knockouts Toxin, oncogene, allergen, modulation of growth-hormone /cytokine and hazards of over expression.
Transfer Impact of dissemination, survival in the environment allowing for gene transfer. More or less hazardous
Is the construct more hazardous than the wild type or less hazardous? Start with the wild type containment measures then determine if additional controls from the Containment level above are required or has the organism been altered in such a way that it can handled with lower controls. Consider the GMO as well as the activity proposed. Environmental hazards
Hazards of: Information required The recipient organism Capability to infect plants, animals, and insects. Can alterations be reverted to wild type. Are the microorganisms included in other legislation e.g., SAPO or Anti-terrorism, crime and security Act, Schedule 5 Inserts and or knockouts Impact on pathogenicity, resistance to therapeutic drugs, or ability to evade host natural defences, are the inserts / knock outs toxins, cytokines or oncogenes Hazards from transfer Impact of widespread dissemination, survival in the environment allowing gene transfer Activity hazards
Hazards of: Information required Aerosol generating activities How will these be managed e.g. MSC / centrifuge buckets / rotors with lids Waste management Specify disinfect procedures, autoclave procedures and back up plans Sharps How will sharps be managed In vivo work Detail the risks of shedding Life cycle of the organisms Is there a hazardous part of the life cycle e.g., parasites where there is an infective stage or release of spores from fungi Are there high-risk users e.g., people who have existing skin conditions e.g., eczema. Immunocompromised or immune suppressed workers Health surveillance requirements Available vaccinations and or health surveillance requirements. Control measures What additional controls are required above those available at the containment level identified. If some activities assigned to different classes, please provide more information on these. - Guidance for Living Genetically Modified Organisms risk assessment
The table below provides guidance on the information required for an GM Animal and GM plant risk assessment.
Factors to consider Information required Risks to the environment if animals escape Stability of modification allowing
Survival – temperature, food sources,
Establishment - out competing, displace or prey on native species, impact on food sources for wild animals
Disseminate genetic modification through breed with wild animals or shedding. Consider selection pressure to revert to wild typeTransfer of genetic material Sexually compatible species, sex and fertility of animals. Female animals are a lower risk to the environment providing they can be retrieved before breeding Insert Does the insert code for any biologically active substances
Does the insert make the animal a disease reservoir e.g., able to express a receptor for virus, or toxinCharacteristics of the proposed activity Detail the proposed activities and the associated risks to human health and the environment Likelihood of adverse effects to human health and the environment How are the animals restricted from accessing the external environment, ease of escape, ease of retrieval of escapees. Consequence of adverse effects to human health and the environment Impact of genetically modified material entering the environment Containment measures Containment and control measures for GM animals
Appropriate size pens / cages / enclosures for the species. There should be physical barriers to prevent escape in addition to the procedure and management controls.Only those activities where the LGMO is more harmful than the parental organism need to be notified to HSE. Work can start 45 days after the date of the acknowledgement or earlier if HSE gives written approval for the work to start.
For further information refer to SACGM Compendium of guidance Part 5 Genetically modification of animals.
- Notifications for Class 2 and Class 3 GMM
All new Class 2 and Class 3 GM work must be notified to the Health and Safety Executive (HSE) (Class 2 and above).
The notification process requires a UCL GM risk assessment in the correct template. After the GMBSSC is satisfied the information in the GM risk assessment is sufficient, an HSE CU 2 form will need to be completed. The word format will be sent out by Safety Services who will assist in submitting HSE notifications. The information on the CU 2 form will appear on the public register.
The HSE also require a payment for notifications. Safety services will request your PTA (Project.Task.Award) codes once you have submitted all the relevant paperwork. Information on HSE fees All notifications to HSE are made via the Safety Services.
Send your CU2 by email to Safety Services - this will then be filed with your assessment on riskNET once we have received either the acknowledgement or consent from HSE.
When can work start?
Class 2 - All new and subsequent Class 2 activities must be notified to HSE. As UCL already has Class 2 activities notified to HSE, work may start on receipt of the acknowledgement from HSE unless they indicate that work should not start. You should receive an acknowledgement within 10 (working) days of receipt of the notification by HSE. This will be filed as an attachment with your assessment in RiskNET
Class 3 - Work may not start without written consent from HSE. This will be granted (or refused) within 90 days of the date of the acknowledgement for the first notification of this activity class, and 45 days for subsequent activities.
Work with transgenic plants or animals - Only those activities where the GMO is more harmful than the parental organism need to be notified to HSE. Work can start 45 days after the date of the acknowledgement or earlier if HSE gives written approval for the work to start
- Class 1 risk assessment approval process
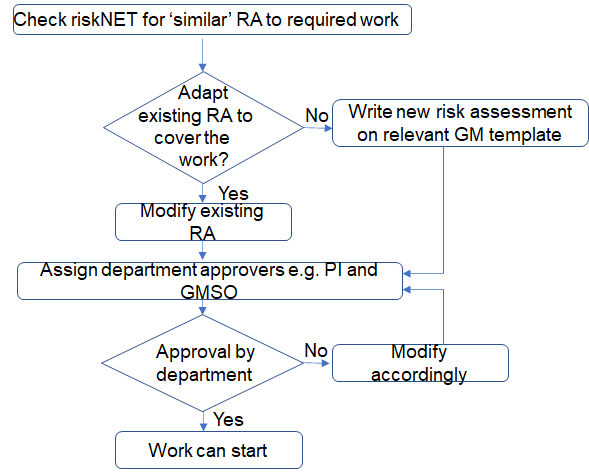
- Class 2 and 3 risk assessment approval process
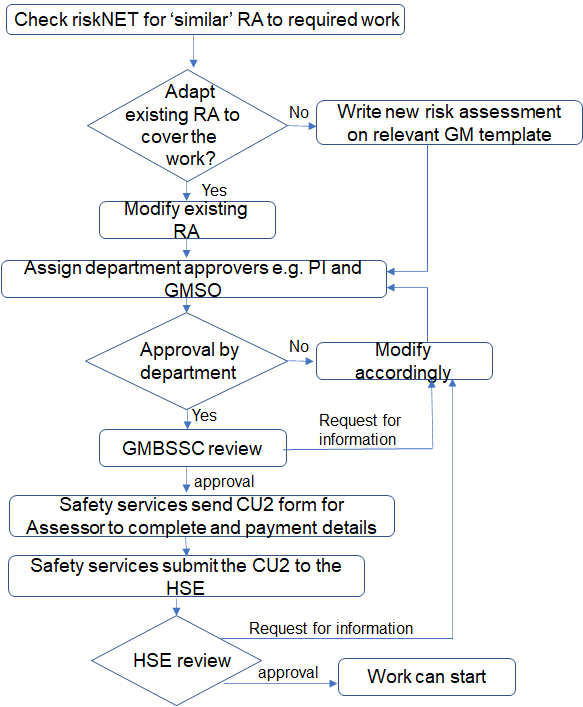
- Guidance on Lentiviral vectors
Class 1 or Class 2?
When classifying any GMO the default is to handle as the wildtype. Then consider if the wild type microorganism has been made more hazardous or less hazardous. This will be determined by the final construct with the inserts and or knock outs. Then the activities involving the GMO have to be considered to determine the final class of the GMO.
The table below provides guidance on when lentiviral vectors can be Class 1 and Class 2.
Class 1 Activity
not notifiable to the HSE
Third generation or above
Contains a Self Inactivating LTR
No harmful inserts
No sharpsClass 2 Activity
notification and a fee required by the HSEUse of first or second generation vector
Include harmful inserts such as cytokines, toxins, oncogenes, growth factors and immunomodulatory proteins.
Use of WPRE which codes of protein X
Use of sharpsFor work to be considered to be class 1 all the criteria listed above must be met. However, for a lentiviral vector to be Class 2 it only needs to meet one criterion listed above or require a single or multiple CL2 controls. The information above should be considered when determining the class of the GM work involving Lentiviral vectors. This will be determined by risk assessment on a case-by-case basis.
The GM risk assessment should also document management and controls when using sharps and aerosol generating activities.
Further information about Lentiviral vectors including information on the generations of Lentiviral vectors can be found in the SACGM compendium of guidance part 2. For further information contact your local GMSO or the Biochem mailbox.
Containment
The containment level is usually based on the hazard group, however, the type of work you are doing may change the containment level required. This should have been assessed during the risk assessment process.
All containment labs and equipment such as microbiological cabinets will require additional department lead training before work can begin.
> Containment Levels
> Storage of GMM
Summary of requirements for approval
Risk assessments must not be written and approved by the same person.
GM risk assessments should be approved by the person in management control of the work, for example, the Principal Investigator. If the PI is the assessor and needs to be listed as an approver, then a peer with knowledge and experience of GM work, such as the local Genetic Modification Safety Officer (GMSO), can approve the risk assessment to confirm the technical detail and the controls to be implemented are suitable
Additional approval for Class 2 and Class 3 Geneticaly Modified material
Once the approvers listed in the GM risk assessment have approved the risk assessment, approval is also required by the GMBSCC before the risk assessment is sent to the HSE for notification for new work or significant changes. Minor amendments Class 2 and Class 3 GM risk assessments that have been notified and agreed by the HSE can be approved by the UCL GMBSSC and will not require further notification unless the scope or the risk has changed.
Class 1 | Class 2 | Class 3 |
Only requires department-level approval | Requires approval by GM working group and BSO | Requires approval by GM working group and BSO |
Safety Services carry out a compliance check once a month | First notification and all reviews to be approved | First notification and all reviews to be approved |
HSE notification Once for the premises All subsequent work covered by the initial notification | HSE notification Required for first contained use HSE must be given 45 days to raise concerns BSO will approve risk assessment after that date if the HSE do not contact UCL Required for subsequent use - once HSE acknowledges notification work can begin after BSO has approved risk assessment | HSE notification Required for first contained use Consent letter will be sent within 90 days On receipt BSO will approve risk assessment Required for subsequent uses Consent letter will be sent within 45 days On receipt BSO will approve risk assessment |
Last updated: Thursday, January 26, 2023
 Close
Close


