The Mineralomics Research Group seeks to answer crucial biological and medical questions related to the nature of biominerals present in different medical contexts.
Overview
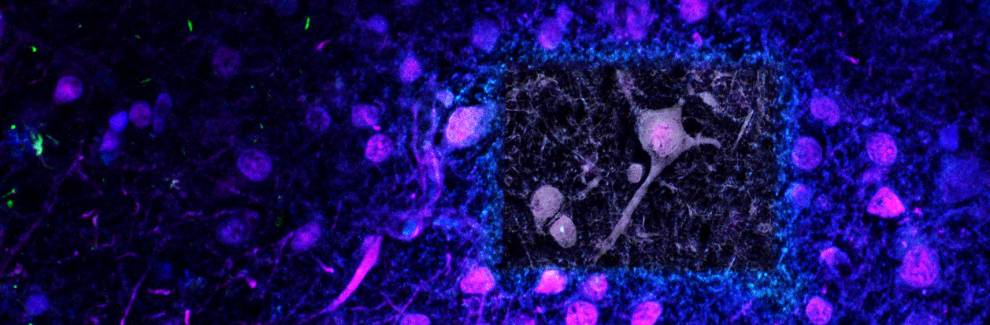
Hard tissues from vertebrates (e.g. bone, teeth and otoliths), invertebrates (e.g. shells and exoskeletons of crustaceans) or calcification in tissues caused by disease such as atherosclerosis and cancer, all share a common feature in that they are all distinctive structures that include both inorganic and organic components. These calcified tissues are all formed from a combination of inorganic minerals and organic proteins and cells. Taken together, they are known as biominerals and are placed at a unique intersection of different fields of study, such as Biology, Medicine, Chemistry and Physics.
Whilst some of the knowledge that we have today about biominerals comes from biomedical research, a large quantity of key information about biominerals has been provided by characterization techniques rooted in physical sciences. Answers to fundamental questions about the nature, origin and function of biominerals have been offered by Electron Microscopy (including Scanning Electron Microscopy [SEM] and Transmission Electron Microscopy [TEM]), diffraction techniques such as x-ray diffractometry and electron diffractometry and, more recently, Focused Ion Beam (FIB).
The Mineralomics research group at UCL Medical Physics & Biomedical Engineering seeks to answer crucial biological and medical questions related to the nature of biominerals present in different medical contexts using and developing several techniques from physical sciences. Working in an interdisciplinary field, our research interests go from natural hard tissues to pathological calcification connected with a wide range of diseases that include the top killers in the world today — from arteriosclerosis and aortic valve stenosis to Alzheimer’s disease and cancers.
The diversity of topics studied in our group reflects the exciting and diverse breadth of mineralomics research. Indeed, the relevance of this fascinating multidisciplinary field is not restricted to the several branches of biology, but can also be seen in disciplines such as medicine, chemistry, geology, materials science and physics.
People
Research Themes
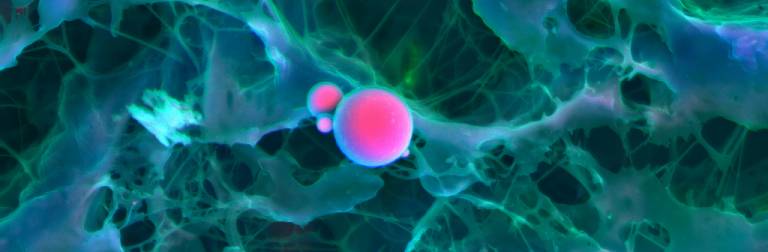
A main focus for our research involves the use of physical-chemistry methods to study the characteristics of biominerals present in calcific disease, the nature of these biominerals, their origin and possible mechanisms of their formation. We hope the outputs of our research will pave the way for new treatments and preventative therapies of diseases as diverse as cancer and atherosclerosis. Our interdisciplinary research has so far considerably improved our understanding of cardiovascular calcification, cancer, Alzheimer’s disease and macular degenerative disease. In 2013, we discovered that the first biominerals found in the vascular system are formed from highly crystalline spherical particles that differ from any other biomineral present in the body. These same minerals are also present on calcification on malign tumours and that a different kind of minerals is present on the nucleus of brain cells.

Selected Publications
- 2020
- https://www.materialstoday.com/biomaterials/comment/mineralomics/
- Tsolaki E, Csincsik L, Xue J, Lengyel I, Bertazzo S (2020) Association of cellular and subcellular calcification with phosphorylated tau in the brains of Alzheimer’s disease patients bioRxiv doi:https://doi.org/10.1101/2020.06.15.148353
- Tsolaki E, Doran W, Magnani L, Olivo A, Herrmann IK, Bertazzo S (2020). Invasive breast tumors are characterized by the presence of crystalline nanoparticles bioRxiv doi:https://doi.org/10.1101/2020.04.29.067660
- Rogers, M. A., Buffolo, F., Schlotter, F., Atkins, S. K., Lee, L. H., Halu, A., . . . Aikawa, E. (2020). Annexin A1-dependent tethering promotes extracellular vesicle aggregation revealed with single-extracellular vesicle analysis. Science advances, 6 (38). doi:10.1126/sciadv.abb1244
- 2019
- Anthis, A. H. C., Tsolaki, E., Didierlaurent, L., Staubli, S., Zboray, R., Neels, A., . . . Herrmann, I. K. (2019). Nano-analytical characterization of endogenous minerals in healthy placental tissue: Mineral distribution, composition and ultrastructure. Analyst, 144 (23), 6850-6857. doi:10.1039/c9an01312a
- Tsolaki, E., & Bertazzo, S. (2019). Pathological Mineralization: The Potential of Mineralomics. Materials (Basel), 12 (19). doi:10.3390/ma1219312
- 2018
- Tan, A. C. S., Pilgrim, M. G., Fearn, S., Bertazzo, S., Tsolaki, E., Morrell, A. P., . . . Curcio, C. A. (2018). Calcified nodules in retinal drusen are associated with disease progression in age-related macular degeneration. Science Translational Medicine, 10 (466), ARTN eaat4544. doi:10.1126/scitranslmed.aat4544
- Wald, D. S., Tsolaki, E., Bestwick, J. P., & Bertazzo, S. (2018). Scanning electron microscopy for blood micro-crystals in aortic stenosis patients. PLoS ONE, 13 (8). doi:10.1371/journal.pone.0202282
- 2017
- van Engeland, N. C. A., Bertazzo, S., Sarathchandra, P., McCormack, A., Bouten, C. V. C., Yacoub, M. H., . . . Latif, N. (2017). Aortic calcified particles modulate valvular endothelial and interstitial cells. Cardiovascular Pahotlogy, 28, 36-45.
- Bertazzo, S., & Gentleman, E. (2017). Aortic valve calcification: a bone of contention. European Heart Journal, 38 (16), 1189.
- You, A. Y. F., Bergholt, M. S., St-Pierre, J. -. P., Kit-Anan, W., Pence, I. J., Chester, A. H., . . . Stevens, M. M. (2017). Raman spectroscopy imaging reveals interplay between atherosclerosis and medial calcification in the human aorta. Science advances, 3 (12), e1701156.
- 2016
- Bertazzo, S. (2016). Cardiovascular calcification and hard sciences: a solid match [Digital scholarly resource]. Retrieved from http://isacb.org/scientific-profile-by-sergio-bertazzo.
- Krohn, J. B., Hutcheson, J. D., Martinez-Martinez, E., Irvin, W. S., Bouten, C. V. C., Bertazzo, S., . . . Aikawa, E. (2016). Discoidin Domain Receptor-1 Regulates Calcific Extracellular Vesicle Release in Vascular Smooth Muscle Cell Fibrocalcific Response via Transforming Growth Factor-beta Signaling. ArteriosclerosisThrombosis and Vascular Biology, 36 (3), 525-533. - Selected for front cover
- Hutcheson, J. D., Goettsch, C., Bertazzo, S., Maldonado, N., Ruiz, J. L., Goh, W., . . . Aikawa, E. (2016). Genesis and growth of extracellular-vesicle-derived microcalcification in atherosclerotic plaques. Nature Materials, 15 (3), 335.
- Agarwal, S., & Bertazzo, S. (2016). New paradigms in cardiovascular calcification. Comptes Rendus Chimie, 19 (11-12), 1605-1609.
- 2015
- Bertazzo, S. (2015). Bio-medical physical sciences – a personal perspective on interdisciplinary research [Digital scholarly resource]. Retrieved from http://www.materialstoday.com/biomaterials/comment/biomedical-physical-s....
- Bertazzo, S. (2015). Biomineralization. Seminars in Cell and Developmental Biology, 46, 1.
- Bazin, D., Jouanneau, C., Bertazzo, S., Sandt, C., Dessombz, A., Réfrégiers, M., . . . Daudon, M. (2015). Combining field effect scanning electron microscopy, deep UV fluorescence, Raman, classical and synchrotron radiation Fourier transform Infra-Red Spectroscopy in the study of crystal-containing kidney biopsies. Comptes Rendus Chimie, 19 (11-12), 1439-1450.
- Kapustin, A. N., Kalinina, N., Lopatina, T., Davidson, S. M., Iraci, N., Tamkovich, S., . . . Tkachuk, V. (2015). UK-Russia researcher links workshop: Extracellular vesicles - mechanisms of biogenesis and roles in disease pathogenesis, M.V. Lomonosov Moscow State University, Moscow, Russia, 1-5 march 2015. Journal of Extracellular Vesicles, 4 (2015), 1-3.
- Kapustin, A. N., Chatrou, M. L. L., Drozdov, I., Zheng, Y., Davidson, S. M., Soong, D., . . . Shanahan, C. M. (2015). Vascular Smooth Muscle Cell Calcification Is Mediated by Regulated Exosome Secretion. Circulation Research, 116 (8), 1312-1323.
- 2014
- Bertazzo, S., Steele, J. A. M., Chester, A. H., Yacoub, M. H. et al (2014). Cardiovascular calcification violet pearl. The Lancet, 384 (9950), 1294.
- Moore, H. M., Willenberg, T., Coleridge-Smith, P., Chiappini, C., Bertazzo, S. et al (2014). The effects of detergent sclerosants on white blood cells. British Journal of Surgery, 101, 29. WILEY-BLACKWELL.
- Chester, A. H., El-Hamamsy, I., Butcher, J. T., Latif, N., Bertazzo, S., & Yacoub, M. H. (2014). The living aortic valve: From molecules to function. Global Cardiology Science and Practice, 2014 (1), 11.
- 2013
- Bertazzo, S., Gentleman, E., Cloyd, K. L., Chester, A. H., Yacoub, M. H. et al (2013). Nano-analytical electron microscopy reveals fundamental insights into human cardiovascular tissue calcification. Nature Materials, 12 (6), 576-583.
 Close
Close

