Nerve cell loss is implicated in chronic and devastating neurodegenerative disorders such as Alzhei-mer's Disease. Although loss of nerve cells in Alzheimer's disease is classically said to occur in specific regions of the brain, as the disease progresses cells all over the central nervous system are affected. Of great interest to eye physicians is the strong association of the loss of nerve cells in the eye with this disease. Moreover, there is increasing evidence that similar mechanisms in the eye and the brain cause the development of this nerve cell loss. This suggests that the processes of cell death occurring in the eye may be an indicator of, or window on, cell death occurring in other parts of the brain.
We have already shown that it is possible to assess therapeutic and sight-saving strategies using our methods and this has great implications in the testing and validation of neuroprotective regimens in all aspects of neurodegeneration including Alzheimers Disease.
Neurodegenerative diseases are characterized by chronic nerve cell loss. Alzheimer's disease is the commonest single cause of dementia. It is an age-dependent disease just like glaucoma, but unlike glaucoma is rare (<1%) in the under-65s. One of the mechanisms that is believed to lead to neurodegeneration in Alzheimer's Disease is the production of a protein called amyloid-beta (Aβ). This protein, which causes the so-called "Alzheimer brains" is only produced when a nerve cell is "stressed". Our recent work has identified the Aβ protein in the retinal nerve cells in glaucoma models, and shown it was associated with nerve cell apoptosis (so-called programmed cell death). Using DARC, we showed that when this Aβ protein was given by itself to "normal" eyes it caused visible nerve cell apoptosis which we were able to monitor in vivo and in real time.
Since Aβ is proven to cause nerve cell death in the retina, we devised a combination therapy to target this protein . Three different drugs were used, each of which act on a different stage in the known Aβ protein production pathway .. One of these drugs is an antibody, like Bapineuzumab, which is already being used in Phase III clinical trials to treat Alzheimer's patients. These treatments targeting the Aβ protein significantly decrease the number of dying retinal nerve cells in glaucomatous eyes. Moreover, when combined with the two other novel Alzheimer's treatments, the effects on glaucoma were even stronger.
The implications for this are wide. Firstly, targeting the Aβ protein opens a brand new avenue of treatment in glaucoma - therapy that is no IOP lowering and therefore of benefit to patients who continue to lose vision despite adequate IOP control. Secondly, the idea of combining therapies working on the same pathway makes biological sense - but has not been done before in either glaucoma or Alzheimer's. Finally, the eye may provide a new "window" on brain disease - one that enables not only diagnosis, non-invasively, but also the monitoring of new neurological treatments.
error message: 'NoneType' object has no attribute 'split'
error message: 'NoneType' object has no attribute 'split'
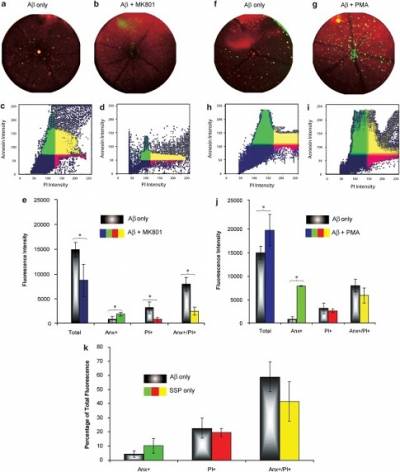
Analysis of cell death phases in a mouse model of Aβ-induced RGC death. (a, b) Wide-angle retinal images showing the Aβ-treated right eye (a) and Aβ+MK801-treated left eye (b) of a single mouse. MK801 reduces the number of annexin V (green)- and PI (red)-positive cells 72h after intravitreal Aβ(25-35) application. To assess the magnitude of late apoptosis, we studied the co-localisation of PI and annexin V using ImageJ software with the Intensity Correlation Analysis plugin. (c, d) Scatter plots show the distribution of annexin and PI fluorescence intensity in the eyes shown in (a, b), Aβ alone (c) and Aβ+MK801 (d). (e) Thresholds were set automatically by the plugin for each analysis (colour overlay in (c, d)), and quantification of each cell death phase was performed from generated data (e). MK801 treatment reduced the overall levels of cell death (blue, total), mostly by reducing the number of cells in late-phase apoptosis (yellow, Anx+/PI+) and necrosis (red, PI+). (f, g) Wide-angle retinal images showing the Aβ-treated right eye (f) and Aβ+PMA-treated left eye (g) of a single mouse. (h, i) PMA increased the level of annexin V (green) and PI (red) positivity, as shown in the corresponding scatter plots. (j) Quantification of all treated animals shows that PMA significantly (P<0.05) increases the overall levels of cell death (blue, total), most of this being due to a significant (P<0.05) increase in the number of cells in the early phase of apoptosis (green, Anx+). (k) Comparison of cell phases in all Aβ and SSP models using co-localisation scatter plot analysis. At the previously established time points of maximal apoptosis (Aβ=72h and SSP=2h), both models show a similar trend, with no significant difference in the magnitude of any phase of cell death. However, both models show significantly more RGCs in the phases of late apoptosis (P>0.05, yellow, Anx+/PI+) and necrosis (P>0.05, red, PI+) than early apoptosis (green, Anx+). Error bars 95% confidence intervals, *P<0.05. From Cell Death Dis. Jan 2010; 1(1): e3.
 Close
Close

