Glaucoma is now a major cause of irreversible blindness worldwide, with a significant socio-economic burden that is predicted to worsen as the population ages.
Visual loss is attributed to degeneration of the retinal ganglion cell (RGC), which is the retinal neuron involved in the transfer of visual information from the eye to the brain.
Unfortunately, conventional clinical tests cannot detect abnormalities until extensive RGC death and significant vision loss have already occurred.
However, it is known that one of the earliest hallmarks of this disease is the process of neuronal "apoptosis" or so-called "programmed" cell death of the RGC.
error message: 'NoneType' object has no attribute 'split'
We have recently devised a novel technique to image and track this process of retinal neurons (RGC) apoptosis in vivo.
DARC is a method by which we can now observe individual nerve cells dying in the living eye.
The strength of this technique is that it may be used as a tool to open a new "window" on to the cellular mechanisms involved in the development of RGC apoptosis and vision loss in glaucoma.
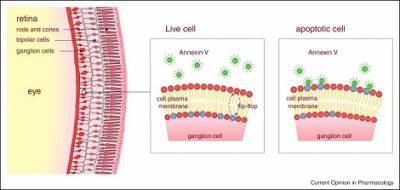
A schematic diagram of the DARC technology. After injection of fluorescent labelled annexin V, apoptotic cells are identified by the binding of annexin to translocated phosphatidylserine to the outer leaflet of the membrane of retinal ganglion cells, which show up as bright white spots in vivo images (Image from Current Opinion in Pharmacology 2013, 13:123-127).
Unique imaging techniques
Our unique imaging techniques open a new "window" on the cellular mechanisms involved in the development of RGC apoptosis and vision loss in glaucoma. Their analysis will enable delineation of signalling pathways that couple glaucomatous disease to neurodegeneration, and should provide an unparalleled platform from which to effectively assess potential neuroprotective strategies.
This project has implications not only in glaucoma and the prevention of blindness, but any condition associated with neuronal apoptosis, where our findings can help enhance the clinical diagnosis and refine therapeutic intervention.
We have been able for the first time to image changes occurring in retinal nerve cells apoptosing over hours, days and months.
This advance will potentially provide a powerful new clinical tool with which to diagnose and identify patients with early glaucoma before they lose vision, and opens the door to directly observing effects of therapeutic strategies in glaucoma using meaningful endpoints that are based on the direct assessment of RGC death.
It may also serve as a surrogate biomarker of outcome in glaucoma clinical studies, dramatically reducing the duration of such trials that currently rely on the use of visual field status as a key endpoint.
Prof. Cordeiro believes it may be possible to have an apoptotic index of cell degeneration with which individuals can be diagnosed and also monitored to assess treatment response or progression rates. This may help negate or avoid glaucomatous visual loss from ever occurring.
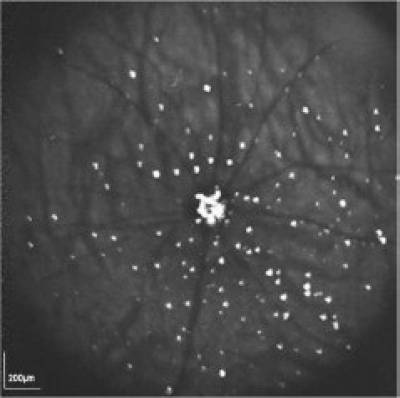
The in vivo retinal image acquired using the DARC (detection of apoptosing retinal cells) technique. The individual apoptosing retinal nerve cells (RGCs, bright spots) in an experimental model of glaucoma are clearly distinguishable. Apoptotic damage was chemically induced in the rat eye 2 hours beforehand. The DARC technique can provide a snapshot of the level of RGC apoptosis at any given timepoint. (Image from Cell Tissue Research 2013)
 Close
Close

