The Frankel Lab
We are part of the UCL Institute of Cardiovascular Science (ICS) within the Research Department of Preclinical and Fundamental Science. Follow us on Twitter (@FrankelLabUCL) for zebrafish, angiogenesis and science news.
Our Research Interests
We are studying angiogenesis (=the formation of blood vessels) in both physiological and pathophysiological situations with an interest in understanding the key signalling pathways required for angiogenesis. Our Group is particularly interested in the role of the Cas family of adaptor proteins P130CAS and NEDD9 and how they function to regulate angiogenesis in development and in cancer. We mainly do this using the zebrafish as a research model - find out more about the exciting zebrafish research across UCL here!
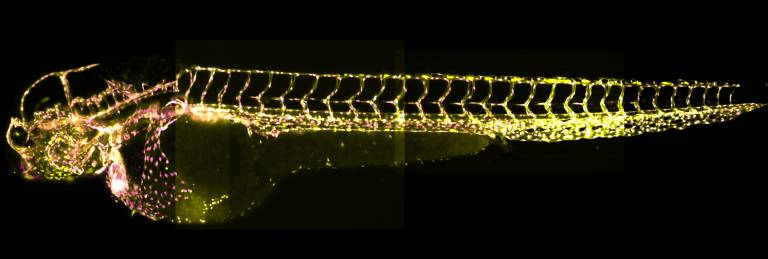
Stitched together image of a 48h old zebrafish embryo we acquired with the Zeiss lightsheet microscope. The embryo has 2 transgenes which label the cytoplasm (=yellow) and nuclei (=magenta) of all endothelial cells in the fish, allowing us to see the shape of the blood vessels and count the number of cells.
The scaffolding protein p130Cas
is a key mediator of angiogenesis and was characterised by work in our department to specifically function downstream of Neuropilin-VEGF signalling in endothelial cells, as well as mediate pro-angiogenic effects in vascular smooth muscle cells. However, not much is understood about its role in vivo beyond a long established requirement for heart organogenesis.
We have generated a novel bcar1-/-(p130Cas) zebrafish line and are using confocal and lightsheet microscopy and a variety of endothelial-specific reporter lines to interrogate p130Cas function. This work is being undertaken by current PhD student Laura Wisniewski.
Furthermore, we have been granted a post-doctoral research position to further analyse the molecular mechanisms through which p130Cas exerts its function, as well as interrogating the role of its closely related family member NEDD9. We are very pleased that Vanessa French will carry out this work.
Tumour angiogenesis
is a critical step in tumour formation as it is required for tumour growth, i.e., like all tissues, tumour cells would not be able to grow without a dedicated blood supply. Tumours are able to actively induce blood vessel growth and the vessels formed are often abnormal, e.g., they are ‘leaky’, twisted in structure, and show disturbed blood flow patterns. This aberrant vasculature is often the primary way that tumours metastasise, as it is easier for cells to enter the blood stream if the endothelium is not intact. Further, the compromised vasculature often complicates anti-cancer treatment, as drugs are not able to reach the entire tumour tisssue.
We have previously shown that two popular drugs, imatinib and nilotinib, actually enhance tumour cell migration through their actions on p130Cas. This work was done by PhD student Tonya Frolov (Read it here). We are following up these observations with a screen to identify the involved kinases and phosphatases.
CS-OPT as a novel imaging modality for vascular development and tumour progression in the adult zebrafish
We have been working closely with the Photonics Group at Imperial College London, with whom we generated a novel imaging platform to investigate vascular development and liver tumourigenesis in adult zebrafish (work done by PhD student Nicola Lockwood, read it here), and with whom we are currently working on longitudinal studies also incorporating drug development and improving imaging technology. This work is being carried out primarily by ICL PhD student Samuel Davis with help from Laura Wisniewski (read it here).
Our current work is supported by grants from the British Heart Foundation and Laura is part of the UCL BHF Fundraising Group - she has previously raised money while doing the NightRide and Prudential50.
last amended 15 February 2019
 Close
Close

