Bowl Lab
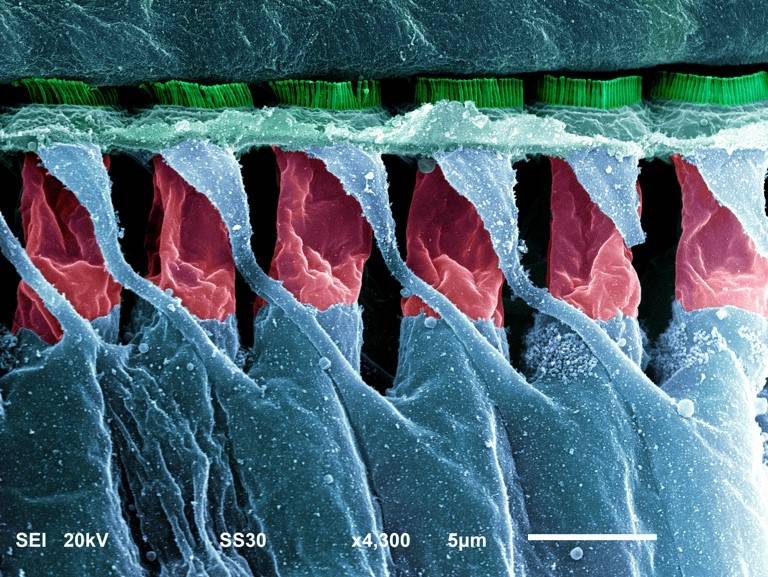
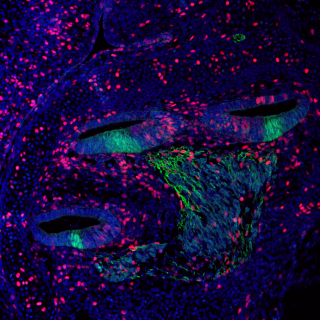
Our research involves utilising the mouse for the identification and study of genes required for mammalian hearing, and the molecular processes underlying hearing loss. Overall, our aim is to increase understanding of the genetic programs involved in cochlear sensory hair cell maturation, function and maintenance, with a particular focus on age-related hearing loss (ARHL), which is a complex disease that is influenced by environmental factors and genetic susceptibility.

A recent Lancet Commission on Dementia identifies midlife peripheral hearing loss as the highest individual risk factor for dementia, and this is pertinent as ARHL is the most prevalent sensory deficit within the population (affects >30% of individuals older than 55-years). However, currently little is known about the genes underlying ARHL. As such, we work closely with collaborators who are undertaking GWAS and exome sequencing screens in ARHL families/cohorts and generate CRISPR/Cas9-mediated mouse models for the highest-ranked genes/alleles arising from these studies. These models allow us to: validate the involvement of specific genes in ARHL causation; identify critical molecular and cellular processes occurring in the aging mammalian ear; determine the pathobiology associated with disease progression (something that is not possible to do in humans due to the inaccessibility of the inner ear); and, assess for mechanistic links underlying cognitive decline associated with peripheral hearing loss. The longer-term view is that increased knowledge will lay the foundations necessary for developing therapeutic strategies to ameliorate progressive hearing loss, and potentially reduce dementia incidence.
Funded by
Current and recent research in the Bowl lab has been supported by the MRC, BBSRC, NIH, Action on Hearing Loss (RNID), and the Dunhill Medical Trust.
 Close
Close

