The study of bacterial epidemiology and evolution has recently undergone a revolution with the advent of next-generation genome sequencing. With the cost of sequencing a complete bacterial genome now comparable to that of a gold-standard typing analysis, traditional molecular methods for investigating bacterial diversity and epidemiology outbreak are increasingly being replaced by genomics. Genomic comparisons of bacterial genomes combined with phylogenetic reconstructions allow performing inferences on the past demographic and evolutionary history of various bacterial species (e.g. their rate of evolution, genomic rearrangements, population sizes, routes of expansions, divergence times). We are interested in applying such tools to unravel the factors allowing certain lineages to outcompete others and boosting their invasive success at various spatio-temporal scales.
•The dynamic of self-transmissible mobile genetic elements in the 7th-pandemic Vibrio Cholerae lineage.
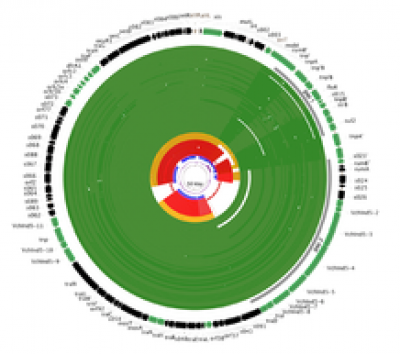
SXT-R391 Integrative conjugative elements (ICEs) are self-transmissible mobile genetic elements able to confer multi-drug resistance and other adaptive features to bacterial hosts, including Vibrio cholerae, the causative agent of cholera. Seven cholera pandemics have been recorded since 1817. The current seventh pandemic that started in 1961 is caused by O1 biotype El Tor, which replaced the classical biotype responsible for previous pandemics. In recent years, several studies using comparative genomics have elucidated the evolutionary relationships of various genetically distinct V. cholera sub-variants. It was for instance demonstrated that they all belong to a single lineage known as the current seventh-pandemic clade that spread from the Bay of Bengal in at least three overlapping waves of strains that shared a common ancestor in the1950s. All those studies however, did exclude ICEs sequences from their evolutionary analyses. ICEs have mostly been studied at an inter-species scale to better understand their organization, regulation and dynamic of transfer. However, little is known about the ICE evolutionary dynamics of a single host species.
In a recent study, we have reported the prevalence, diversity, and dynamics of recombination within the SXT-R391 family, as well as their appearance and spread in the Seventh-pandemic V. cholera lineage. Using comparative genomic analyses we first demonstrated that homologous recombination is an important process shaping the current ICE diversity in the V. cholera seventh-pandemic lineage. In a second step, by carefully selecting some non-recombining ICEs sequences, we were able to perform tip-dating phylogenetic inferences and to date the independent ICEs acquisition by ICE-free V. cholera strains. The concordance between dates of such acquisitions and the strains replacement in between the 2nd and the 3rd wave of transmission illustrate the importance of both ICEs transfer and subsequent recombination in shaping the current epidemiologic dynamic of the disease.
 Close
Close

