UCL is a leading centre for biomedical research in the UK. Scientific research is conducted not by shadowy figures in ivory towers, but by human beings working earnestly to address major issues facing society today.

Dr Clare Stanford: using mice to find treatments for ADHD
Dr Clare Stanford is a Reader in Experimental Psychopharmacology at UCL. Despite the intimidating title, Clare is a down-to-earth, compassionate researcher with a real commitment to animal welfare. She is chair of the Bloomsbury AWERB and does not hold back from questioning the ethics of research objectives, as well as the way it is carried out.
Clare is currently working on a mouse model for Attention Deficit Hyperactivity Disorder (ADHD). This is a strongly inherited psychiatric disorder, which causes problems for patients by making them hyperactive, excessively impulsive and inattentive. ADHD is often regarded as a childhood issue, but about 65% of people carry it through to adulthood where the associated problems are far worse. It has been associated with alcohol and drug misuse in later life, and an estimated 25% of the prison population have ADHD. There is also an increased risk of other health complications, including asthma and epilepsy.
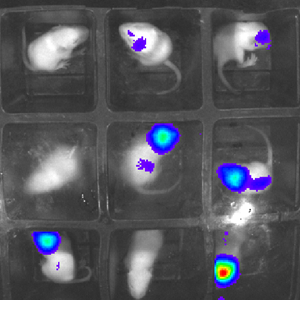
Dr Simon Waddington and Rajvinder Karda: reducing mouse use with glowing firefly genes
Although animal research remains a necessary part of modern research, current methods are far from perfect. By injecting the genes that fireflies use to emit light into newborn mice, UCL scientists have developed a way to drastically reduce the numbers of mice needed for research into disease and development.
At the moment, researchers often need to cull and perform autopsies on animals to see how diseases develop on a molecular level. This means that an animal needs to be killed for every data point recorded, so some studies might use dozens of mice to get reliable data on disease progression.
The new technique could allow researchers to get molecular-level data by simply taking a picture with specialist equipment rather than killing an animal, allowing them to get data more regularly and ethically. An experiment that previously need 60 mice can be done with around 15, and the results are more reliable.

Dr Karin Tuschl: Using zebrafish to treat a rare form of childhood Parkinsonism
Using genetically modified zebrafish, UCL scientists have identified a novel gene affected in a devastating disorder with childhood-onset Parkinsonism. Indeed, when a drug that worked in the fish was given to one of the children, she regained the ability to walk.
The research studied a group of nine children who suffered from severely disabling neurological symptoms including difficulties in walking and talking. Dr Karin Tuschl and her team at the UCL Great Ormond Street Institute of Child Health and UCL Department of Cell and Developmental Biology used state of the art genome editing in zebrafish to validate the identity of the gene affected in these children.
The scientists disrupted a gene known as slc39a14 in the fish, which is important for transporting metals in the body. Disrupting the transporter in fish led to a build up of manganese in the brain and impaired motor behaviour. As similar symptoms were seen in the patients, this confirmed that slc39A14 is required to clear manganese from the body and protect it from manganese toxicity. It also confirmed that the scientists had found the
gene causing the disease in the patients.
 Close
Close

