Mobile Decision Support Tool to Enable Effective Decentralised Management of Drug-Resistant TB
A mobile decision support tool to enable effective decentralised management of drug-resistant Tuberculosis in adults and children.
16 March 2020
The Challenge for the Research
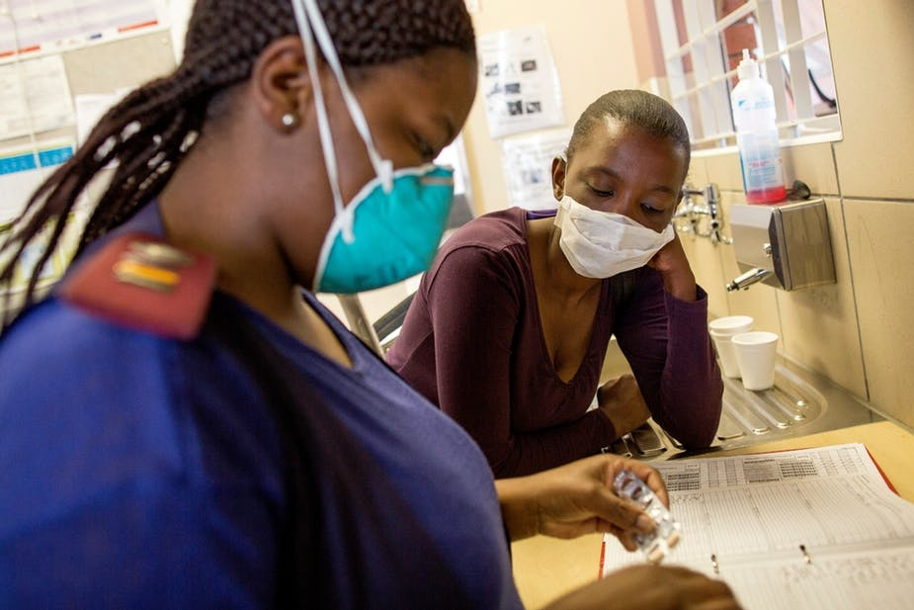
Drug-resistant (DR) tuberculosis (TB) is the most costly and complex form of TB to manage. In 2017 there were estimated to be 558,000 cases of DR-TB globally and 230,000 deaths. The 30 countries which account for more than 90% of these cases are all ODA eligible countries. South Africa has the 3rd highest number of notified cases of DR-TB and high rates of HIV co-infection. Treatment for DR-TB can take up to 20 months and require concurrent administration of up to 7 drugs which require careful toxicity monitoring. DR-TB has therefore often only been managed at small numbers of specialist hospitals where expertise is concentrated. This contributes to a case of DR-TB costing 6-times that of DS-TB to manage, in South Africa DR-TB consumes 65% of the total TB budget while representing only 7% of cases.
Recent successful pilots in South Africa of decentralised management, initiating and following up DR-TB cases in primary care and non-specialist inpatient facilities rather than specialist hospitals, have shown considerably reduced costs and expanded access. This approach has been shown to reduce cost by 74% with equivalent outcomes while widening access to care and reducing time to treatment initiation.
New guidelines for DR-TB have increased the complexity of management which is a barrier to implementation of the decentralised model of care. Junior healthcare workers frequently seek informal advice via email or WhatsApp groups as smartphone penetration is high. To successfully implement an effective decentralised model of care, primary care practitioners requires additional support in decision making particularly for paediatric cases and HIV co-infection. Much of the clinical decision making is algorithmic that lends itself well to a digital approach embedded within a smartphone application to assist choice of initial regiment, toxicity monitoring and initial management of common complications.
Project aims
- To develop and pilot a smartphone application that provides clinical decision-making support for inexperienced healthcare workers managing drug-resistant TB in adults and children with or without HIV co-infection in decentralised settings in South Africa
- To establish a network to allow future evaluation and implementation of this smartphone application in South Africa and other ODA eligible countries.
Planned approach
We will utilise the complementary experience and expertise of this cross-disciplinary team of TB clinicians from UK and South Africa and the dPHE team at UCL to develop the smartphone application taking the following steps.
- The South African guidelines for DR-TB (2019) will form the basis of the decision tree algorithm. The clinical team will define the minimum data requirement and key decision points to construct an algorithm for initial regimen selection and dosing of DR-TB regimen for children and adults with and without HIV and define the content library for the application.
- To fully understand information needs and existing barriers and facilitators to guideline adherence, we will carry out an online survey followed by a series of focus groups with stakeholders and practitioners in South Africa. Behaviour change theory will be used to structure survey items / interview schedules for the groups to help identify barriers and facilitators to compliance and good practice.
- Working through a co-design process with local healthcare providers, we will integrate the developed decision tree algorithm into design of a prototype wireframe and range of mock-up pages for the application. Pages will integrate positive and negative feedback as well as a range of gamified features (e.g. badges, points system, interactive mentor) to promote sustained engagement, to reinforce learning and to encourage sustained change to DR-TB decision making behaviours. Following an iterative testing phase to refine application structure, features and feedback text, a prototype of the app will be developed. The developed tool will be tested with a range of hypothetical scenarios in HIV infected and uninfected adults and children and evaluated for clinical accuracy and usability. It will be demonstrated in 3 decentralised settings to end-users, policy makers and implementation partners in South Africa prior to initial piloting.
Project Funders and Investigators
This project is funded by UCL GCRF Small Grants Fund and brings together teams from across disciplines of computer science, behavioural science, infection control, medicine and global health.
Principal Investigators:
Dr. Hanif Esmail, UCL Institute for Global Health
Dr Patty Kostkova, UCL IRDR Centre for Digital Public Health in Emergencies
Project team:
Dr Caroline Wood, UCL IRDR Centre for Digital Public Health in Emergencies
Dr. Bianca Sossen, UCL Institute for Global Health
Georgiana Birjovanu, UCL IRDR Centre for Digital Public Health in Emergencies
Dr. Susanne Luedtke, UCL IRDR Centre for Digital Public Health in Emergencies
Partners:
Dr. Jennifer Hughes, University of Stellenbosch SA
Dr. Tom Boyles, University of Witwatersrand SA
Dr. Adam Gray, University College London Hospital NHS Trust
Period of Award
January – June 2020
 Close
Close

