First comprehensive map of human blood stem cell development
13 April 2022
A first-of-its-kind roadmap traces each step in the development of blood stem cells in the human embryo, providing scientists with a blueprint for producing fully functional blood stem cells in the lab, reports a team led by UCL and UCLA scientists.
The research, published today in Nature, could help expand treatment options for blood cancers like leukaemia and inherited blood disorders such as sickle cell disease.
Blood stem cells, also called hematopoietic stem cells, have the ability to make unlimited copies of themselves and to differentiate into every type of blood cell in the human body. For decades, doctors have used blood stem cells from the bone marrow of donors and the umbilical cords of newborns in life-saving transplant treatments for blood and immune diseases. However, these treatments are limited by a shortage of matched donors and hampered by the low number of stem cells in cord blood.
Researchers have sought to overcome these limitations by attempting to create blood stem cells in the lab from human pluripotent stem cells, which can potentially give rise to any cell type in the body. But success has been elusive, in part because scientists have lacked the instructions to make lab-grown cells differentiate into self-renewing blood stem cells rather than short-lived blood progenitor cells, which can only produce limited blood cell types.
Co-corresponding author of the research, Dr Vincenzo Calvanese (UCL Laboratory for Molecular Cell Biology), who began the research at UCLA before moving to UCL and completing the analysis, said: “Previous efforts to make functional blood stem cells in the lab have not been successful as we did not know enough about how these cells develop. Our new roadmap will help other scientists to understand fundamental differences between blood stem cells and short-lived progenitor cells.
“We now have a manual of how hematopoietic stem cells are made in the embryo and how they acquire the unique properties that make them useful for patients.”
The research team, which also included scientists from Germany’s University of Tübingen and Australia’s Murdoch Children’s Research Institute, created the resource using single-cell RNA sequencing and spatial transcriptomics, new technologies that enable scientists to identify the unique genetic networks and functions of thousands of individual cells and to reveal the location of these cells in the embryo.
The de-identified data are available to the public on the website The Atlas of Human Hematopoietic Stem Cell Development.
The data make it possible to follow blood stem cells as they emerge from the hemogenic endothelium and migrate through various locations during their development, starting from the aorta and ultimately arriving in the bone marrow. Importantly, the map unveils specific milestones in their maturation process, including their arrival in the liver, where they acquire the special abilities of blood stem cells. The researchers explain that immature blood stem cells must move through different locations to learn how to do their job as blood stem cells.
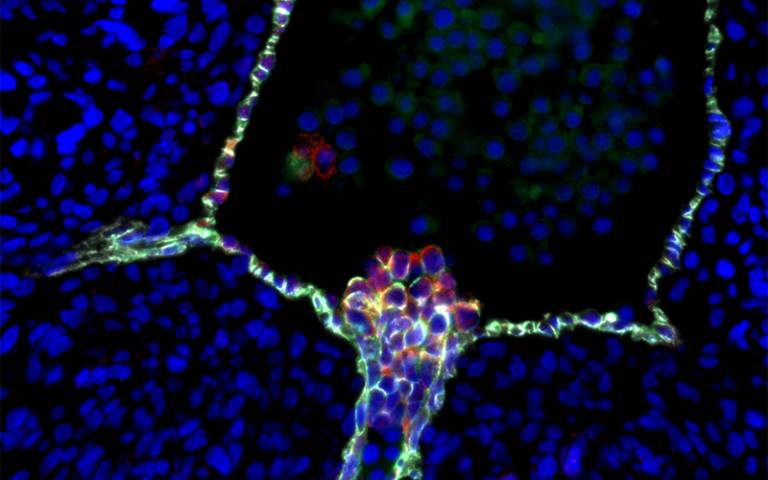
The research group also pinpointed the exact precursor in the blood vessel wall that gives rise to blood stem cells. This discovery clarifies a longstanding controversy about the stem cells’ cellular origin and the environment that is needed to make a blood stem cell rather than a blood progenitor cell.
Now that the researchers have identified specific molecular signatures associated with the different phases of human blood stem cell development, scientists can use this resource to see how close they are to making a transplantable blood stem cell in the lab.
Lead author Professor Hanna Mikkola (Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA) said: “Previously, if we tried to create a blood stem cell from a pluripotent cell and it didn’t transplant, we wouldn’t know where in the process we failed.
“Now, we can place the cells in our roadmap to see where we’re succeeding, where we’re falling short and fine-tune the differentiation process according to the instructions from the embryo.”
In addition, the map can help scientists understand how blood-forming cells that develop in the embryo contribute to human disease. For example, it provides the foundation for studying why some blood cancers that begin in utero are more aggressive than those that occur after birth.
Professor Mikkola added: “Now that we’ve created an online resource that scientists around the world can use to guide their research, the real work is starting. It’s a really exciting time to be in the field because we’re finally going to be seeing the fruits of our labour.”
The research was supported by the National Institutes of Health, the UCLA Jonsson Cancer Center Foundation, the David Geffen School of Medicine at UCLA, the Swedish Research Council, the European Molecular Biology Organization, the Swiss National Science Foundation and the UCLA Broad Stem Cell Research Center (including support from the Rose Hills Foundation and the centre’s training program).
Links
- Research paper in Nature
- The Atlas of Human Hematopoietic Stem Cell Development
- Dr Vincenzo Calvanese’s academic profile
- UCL Laboratory for Molecular Cell Biology
Source
- UCLA
Image
- Human blood stem cells emerging from specialised endothelial cells in the wall of an embryonic aorta. UCLA scientists’ confirmation of this process clarifies a longstanding controversy about the stem cells’ cellular origin. Credit: Hanna Mikkola Lab/UCLA, Katja Schenke-Layland Lab/University of Tübingen, Nature
Media contact
Chris Lane
tel: +44 20 7679 9222
E: chris.lane [at] ucl.ac.uk
 Close
Close

