Muscle regeneration secret unlocked
16 August 2005
A research team from UCL's Eastman Dental Institute (EDI) and the Medical Research Council Clinical Sciences Centre has found the cell responsible for muscle repair.
 The discovery has huge implications for treatment of muscle-wasting diseases such as muscular dystrophy.
The discovery has huge implications for treatment of muscle-wasting diseases such as muscular dystrophy.
The findings, published in the journal 'Cell', conclusively demonstrated that satellite cells - the cells that coat muscles - are stem cells that have the ability to produce large quantities of new muscle and which also renew themselves.
Muscle is the most abundant tissue in the body and is responsible for movement, maintaining body temperature and metabolism, as well as providing the body's main store of energy and protein. Decline in muscle is a major problem for the elderly and for many cancer and other patients, as well as those with muscle diseases, and can lead to severe disability or even death. This new discovery has revealed the mechanisms behind muscle regeneration and could lead to novel therapies for muscle-wasting conditions.
It has long been suspected that satellite cells are responsible for muscle regeneration, but this has never been proved until now. The team grafted a very small amount of satellite cells onto muscle tissue, then measured the amount of new muscle generated and the new satellite cells. Each grafted cell produced a large amount of new muscle, and the satellite cells also replicated themselves and formed many new satellite cells.
Professor Irwin Olsen (EDI) said: "This research shows, for the first time, that in a normally stable tissue such as muscle, these stem cells have the ability to produce large quantities of new muscle and also to renew themselves in vivo. Such cells are likely to be fundamental in muscle repair and regeneration following injury and damage by diseases such as muscular dystrophy."
To find out more, use the links at the top of this article.
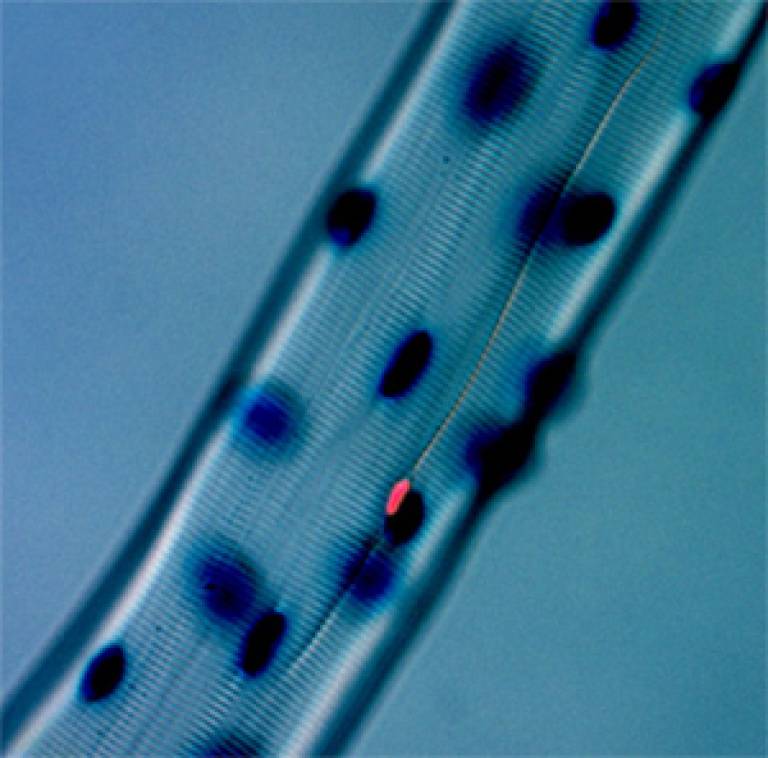
Image: A single myofibre isolated from a muscle. Each mature myofibre contains several hundred nuclei, which are here stained blue. Associated with the myofibre is a 'satellite' cell, with the nucleus stained pink. The team have shown that such satellite cells can function as true muscle stem cells, being able to proliferate and generate large numbers of new myofibres and also to make many more new satellite cells.
Links:
 Close
Close

