Group Leader: Professor Kerry Chester
Introduction
Our research goal is to generate original antibody-based medicines for imaging and therapy of cancer. Our focus is on design and construction of the molecules and their interaction with cancer targets. The basic antibody fragment we use is a single chain Fv (scFv) [Fig 1] and our primary cancer target is carcinoembryonic antigen (CEA), a glycoprotein expressed in colorectal adenocarcinomas. MFE-23, our most characterised scFv, reacts with CEA and was the first scFv to target cancers in man. Our other targets include breast and prostate cancer and the avß6 integrin which is present on head and neck cancers.
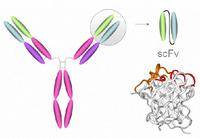
Fig 1 IgG (150 kD) and scFv (27kD). For scFv, antibody VH and VL chains are genetically tethered with a flexible linker to produce a single polypeptide chain.
We obtain the cancer-reactive scFvs from filamentous phage libraries that contain many millions of diverse antibody specificities and we engineer the scFvs in a format designed or a particular therapy. For example, antibody-directed enzyme-prodrug therapy (ADEPT) of cancer, a treatment that uses a systemically administered anti-tumour antibody-enzyme complex to localize enzyme in tumours. In a second stage, a prodrug is administered and is selectively converted into an active cytotoxic drug by enzyme at the tumour site. [Fig 2].

Fig 2 ScFv targets enzyme (blue) to antigen on cancer cell (red). Targeted enzyme subsequently catalyzes prodrug into active cytotoxic (green). The toxic drug is thereby restricted to cancerous areas and does not harm healthy organs.
ADEPT has the potential to generate high concentrations of cytotoxic agent selectively within tumours but it is key for success that non-tumour associated enzyme is effectively cleared from blood and other normal tissues before prodrug administration. A recombinant fusion protein, comprising MFE-23 fused to the enzyme carboxypeptidase G2 (CPG2), has been designed and produced by the group to meet this challenge [Sharma et al 2005, Kogelberg et al 2006].
The therapeutic protein is expressed and purified from yeast Pichia pastoris, and the post-translational glycosylation added by this organism is being exploited to control blood clearance via the mannose receptor [Fig 3] and modify bio-distribution. The therapeutic system is effective in pre-clinical tests [Sharma et al., 2006] and is currently in Phase I/II Clinical Trials [Mayer et al., 2006].
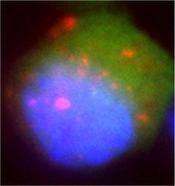
Fig 3 Indirect fluorescence confocal microscopy of single cell (cytoplasm green, nucleus blue) showing MFECP fusion protein (pink) internalized by human mannose receptor.

The strong translational theme of the group is central to our research thinking. The therapeutics designed by the team can be brought to clinical trial because the group have a dedicated facility which can produce clinical grade microbially-expressed recombinant proteins in compliance with Good Manufacturing Practice (GMP) [Fig 4] [Tolner et al, 2006 a,b;].
Fig 4. Making recombinant therapeutics in GMP facility.
The Group are also developing new molecular formats for radioimmunotherapy (RIT) and drug delivery and we are exploring a series of scFv fusion proteins with human serum albumin (HSAbodies). These molecules are designed to be multivalent for antigen, have controllable pharmacokinetics and avoid the human immune system. We have shown that HSAbodies have specific tumour uptake and retention [Fig 5] and that there is little kidney accumulation of radio-labelled material during elimination, which is particularly important for RIT.
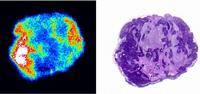
Fig 5 In vivo localisation of 131I HSAbody to viable regions of human tumour xenografts, 24hr post injection. Phosphor image (LHS) and H&E of tumour sections.
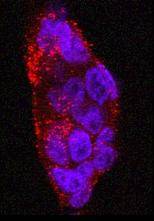
We have recently been using scFv molecules to target iron oxide magnetic nanoparticles [Fig 6] which could be used for specific cancer imaging using MRI. A therapeutic application, by creating localized hyperthermia of cancerous tissue upon application of an alternating magnetic field to targeted nanoparticles is also being explored.
Fig 6 Specific localisation of scFv-functionalized magnetic particles to human cancer cells in vitro.
Selected publications
Schumacher FF, Sanchania VA, Tolner B, Wright ZV, Ryan CP, Smith ME, Ward JM, Caddick S, Kay CW, Aeppli G, Chester KA and Baker JR (2013) Homogeneous antibody fragment conjugation by disulfide bridging introduces 'spinostics'. Scientific Reports 3:1525 Pubmed
Goldstein R, Sosabowski J, Vigor K, Chester K and Meyer T. (2013) Developments in single photon emission computed tomography and PET-based HER2 molecular imaging for breast cancer.Expert Rev Anticancer Ther 13:359-73. Pubmed
Marquardt JL Jr, Begent RH, Chester K, Huston JS, Bradbury A, Scott J, Thorpe PE, Veldman T, Reichert JM and Weiner LM (2012) IBC's 23rd Antibody Engineering and 10th Antibody Therapeutics Conferences and the Annual Meeting of The Antibody Society: December 2-6, 2012, San Diego, CA. MAbs 24;4(6). Pubmed
Chen C, Constantinou A, Chester KA, Vyas B, Canis K, Haslam SM, Dell A, Epenetos AA and Deonarain MP. (2012) Glycoengineering approach to half-life extension of recombinant biotherapeutics. Bioconjug Chem 23:1524-33. Pubmed
Adair JR, Howard PW, Hartley JA, Williams DG and Chester KA. (2012) Antibody-drug conjugates - a perfect synergy. Expert Opin Biol Ther 12:1191-206, review. Pubmed
Miller J, Doss M, McQuillen R, Shaller CC, Tolner B, Yu JQ, Chester K and Robinson MK. (2012) Impact of expression system on the function of the C6.5 diabody PET radiotracer. Tumour Biol 2012 33:617-27Pubmed
Tolner B, Bhavsar G, Foster B, Vigor K and Chester KA (2012) Chapter-37: Production of Recombinant Proteins from Pichia pastoris: Interfacing Fermentation and Immobilized Metal Ion Affinity Chromatography, In: Laboratory Protocols in Fungal Biology; (Eds.) Gupta VK, Tuohy M, Manimaran A, Turner K and Ovonvan A. Springer Science & Business Media, LLC, 233 Spring Street, New York, NY 10013, USA.
Andrady C, Sharma SK, Chester KA (2011) Antibody-enzyme fusion proteins for cancer therapy. Immunotherapy 3:193-211. Review. Pubmed
Marsh D, Suchak K, Moutasim KA, Vallath S, Hopper C, Jerjes W, Upile T, Kalavrezos N, Violette SM, Weinreb PH, Chester KA, Chana JS, Marshall JF, Hart IR, Hackshaw AK, Piper K, Thomas GJ (2011) Stromal features are predictive of disease mortality in oral cancer patients. J Pathol 223:470-81. Pubmed
Vigor KL, Kyrtatos PG, Minogue S, Al-Jamal KT, Kogelberg H, Tolner B, Kostarelos K, Begent RH, Pankhurst QA, Lythgoe MF, Chester KA (2010) Nanoparticles functionalized with recombinant single chain Fv antibody fragments (scFv) for the magnetic resonance imaging of cancer cells. Biomaterials 31:1307-15. Pubmed
Meyer T, Gaya AM, Dancey G, Stratford MR, Othman S, Sharma SK, Wellsted D, Taylor NJ, Stirling JJ, Poupard L, Folkes LK, Chan PS, Pedley RB, Chester KA, Owen K, Violet JA, Malaroda A, Green AJ, Buscombe J, Padhani AR, Rustin GJ, Begent RH (2009) A phase I trial of radioimmunotherapy with 131I-A5B7 anti-CEA antibody in combination with combretastatin-A4-phosphate in advanced gastrointestinal carcinomas. Clin Cancer Res 15:4484-92. Pubmed
Constantinou A, Epenetos AA, Hreczuk-Hirst D, Jain S, Wright M, Chester KA, Deonarain MP (2009) Site-specific polysialylation of an antitumor single-chain Fv fragment. Bioconjug Chem 20:924-31. Pubmed
Yong M, Tolner B, Nagl S, Pedley RB, Chester K, Green AJ, Mayer A, Sharma S, Begent R (2009) Data standards for minimum information collection for antibody therapy experiments. Protein Eng Des Sel 22:221-4. Pubmed
Kogelberg H, Tolner B, Sharma SK, Lowdell MW, Qureshi U, Robson M, Hillyer T, Pedley RB, Vervecken W, Contreras R, Begent RHJ, Chester KA (2006) Clearance Mechanism of Mannosylated Antibody-Enzyme Fusion Protein used in Experimental Cancer Therapy. Glycobiology 17: 36-45. Pubmed
Mayer A, Francis RJ, Sharma SK, Tolner B, Springer CJS, Martin J, Boxer GM, Bell J, Green AJ, Hartley JA, Cruickshank C, Wren J, Chester KA, Begent RHJ (2006). A Phase I Study of Single Administration of Antibody-Directed Enzyme Prodrug Therapy with the Recombinant Anti-Carcinoembryonic Antigen Antibody-Enzyme Fusion Protein MFECP1 and a Bis-Iodo Phenol Mustard Prodrug. Clin Cancer Res.12: 6509-16. Pubmed
Tolner B, Smith L, Begent RHJ & Chester KA. (2006a) Production of Recombinant Protein in Picha pastoris by Fermentation. Nature Protocols 1:1006-1021.
Tolner B, Smith L, Begent RHJ & Chester KA. (2006b) Expanded Bed Adsorption Immobilized Metal Affinity Chromatography. Nature Protocols 1:1213–1222.
Sainz-Pastor N, Tolner B, Huhalov A , Kogelberg H, Lee YC, Zhu D, Begent RHJ, Chester KA (2005) Deglycosylation to obtain stable and homogeneous Pichia pastoris–expressed N-A1 Domains of Carcinoembryonic Antigen. International Journal of Biological Macromolecules 39: 141-150. Pubmed
Sharma SK, Pedley RB, Bhatia J, Boxer GM, El Emir E, Qureshi U, Tolner B, Lowe H, Michael NP, Minton N, Begent RH, Chester KA (2005) Sustained tumor regression of human colorectal cancer xenografts using a multifunctional mannosylated fusion protein in antibody-directed enzyme prodrug therapy. Clin Cancer Res 11: 814-825. Pubmed
Chester K, Pedley B, Tolner B, Violet J, Mayer A, Sharma S, Boxer G, Green A, Nagl S, Begent R (2004) Engineering antibodies for clinical applications in cancer Tumour Biol 25: 91-98. Pubmed
 Close
Close

