Transcription factors play the ultimate role in dictating cell identity, but we still don’t fully understand the molecular basis of their function. Differentiation of naïve CD4+ T cells into different helper subtypes allows tailoring of the immune response to different pathogens and to cancer. However, abnormal patterns of T cell polarization can lead to autoimmune and inflammatory diseases or allergies and T cell dysfunction limits the immune response to cancer. This provides a powerful model system for the study of transcription factor function in cell specification and disease.
The lineage-determining transcription factors T-bet and GATA3 control Th1 and Th2 differentiation, respectively. T-bet directly activates the Th1 signature cytokine interferon gamma (IFNG) and represses the Th2 cytokines IL4, IL5 and IL13, while Gata3 displays the opposite functionality. We have extended this knowledge of T-bet and GATA3 function by showing that the proteins bind across extensive cis-regulatory regions at a key set of immune regulatory genes (Kanhere et al., 2012; Gokmen et al., 2013; Henderson et al., 2021). We have also found that T-bet binding to its target genes is disrupted by genetic variants that are associated with inflammatory disease suggesting that alterations in T-bet function may be a causative factor in these cases (Soderquest et al., 2017).
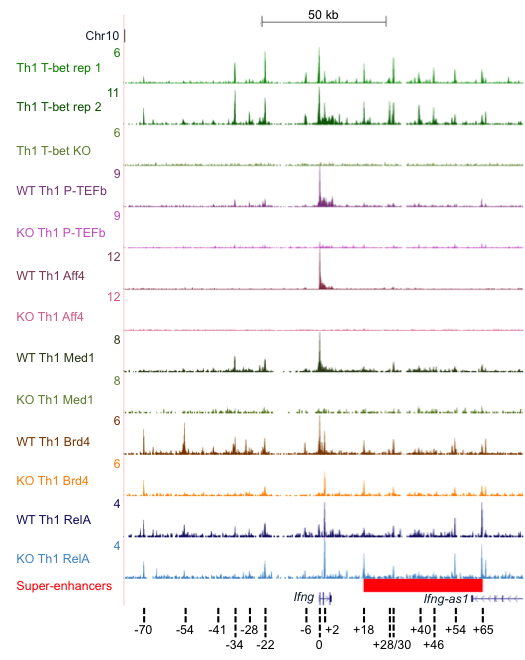
We are also investigating the mechanisms through which T-bet activates Th1 gene expression. We have found that T-bet activates genes through recruitment of the elongation factor P-TEFb (Hertweck et al., 2016; Figure 1) and that CDK9 inhibitors that block P-TEFb function abrogate Th1-mediated inflammatory disease (Hertweck et al., 2016). We are currently seeking to understand the mechanisms through which T-bet recruits co-activators and how its interplay with other lineage-determining transcription factors governs CD4+ T cell fate. We have recently extended our studies to tumour-infiltrating T cells. With Sergio Quezada, we determined the contributions of Blimp-1 and T-bet to cytotoxic CD4+ T cell function in tumour immunity (Śledzińska et al., 2020) and we are currently identifying the mechanisms through which these transcriptional regulators mediate their effects.
 Close
Close


