New insight into the effect of mass transfer on the synthesis of silver and gold nanoparticles
26 March 2019
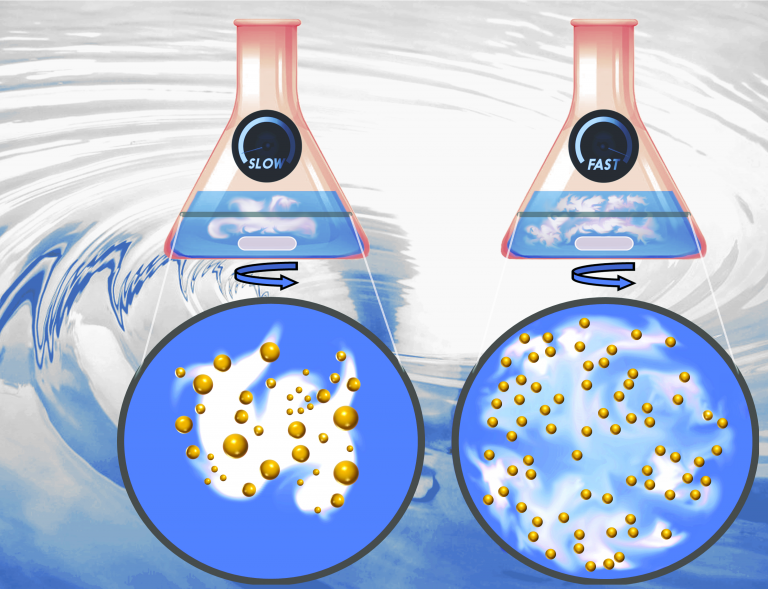
The large number of studies on the silver nitrate reduction synthesis by sodium borohydride for silver nanoparticles and the Turkevich method for gold nanoparticles provide an advanced understanding of the mechanisms of these two syntheses and offers several options to control the NP size distribution. Nevertheless, there is a lack of studies which focus specifically on mass transfer and how it affects the resultant NP size distribution.
This study investigates these two commonly used methods with a focus on the role of mass transfer in the resultant size and polydispersity of the respective NP system. This work highlights the importance of mixing conditions and the order of reagent addition in determining the size and polydispersity of the synthesized NPs, even when the final nominal concentrations of the reducing agent to precursor are the same. Often, batch reactor studies state only the molar ratio of the reagents used in the syntheses, but this is not necessarily sufficient information to reproduce results (explaining the common difficulties of reproducing synthesis in different laboratories), since the way reagents are mixed plays a crucial role.
 Close
Close

