New publication in Nature Microbiology for Banerjee Lab
24 July 2017
Biphasic growth dynamics control cell division in Caulobacter crescentus
Authors: Shiladitya Banerjee, Klevin Lo, Matthew K. Daddysman, Alan Selewa, Thomas Kuntz, Aaron R. Dinner & Norbert F. Scherer.
Cell size is specific to each species and impacts cell function. How cells regulate and sense their size poses a fundamental question in cell biology. Cells precisely control their size during cell cycle to achieve a narrow size distribution within a population. Furthermore, cells can adapt their size and shapes in response to environmental variations, thereby translating extracellular information into decisions to grow or divide. Recent technological advances in single cell imaging have yielded unprecedented amounts of quantitative information about the shapes of single cells as they grow and divide. Based on single-cell experiments, various phenomenological models of size control have been proposed. The "timer" model proposes that newborn cells precisely regulate the amount of time they grow before dividing. However, this model is inconsistent with the observed negative correlation between cell size and division timing.
Recent data on E.coli, B. subtilis, and other bacterial species have argued against a precise control of absolute cell size or division timing. Instead, it has been shown that single bacterial cells follow an "adder" mechanism of size control, whereby they add a constant volume of growth irrespective of their size at birth. However, the bacterial cell cycle is composed of various coupled processes, including DNA replication and cell wall constriction, that have to be faithfully coordinated for cells to divide successfully. This raises the question of what cell cycle variables regulate the added cell size and how cell shape changes act as sensors for cell size and division control.
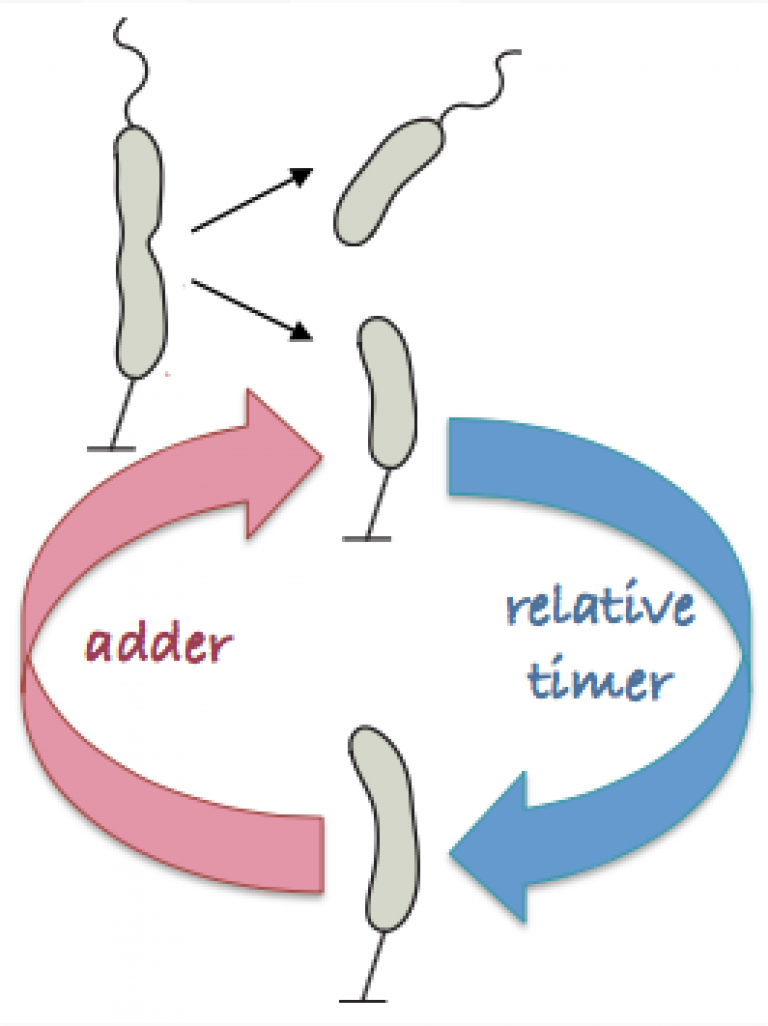
In this study, Banerjee and coworkers at the University of Chicago show that rod-like bacteria follow a two-step mechanism of growth that combines a "timer" and an "adder" model. Using Caulobacter crescentusas a model organism, they show that C. crescentus cells grow for a fixed amount of time relative to the cell cycle before they initiate constriction of the cell wall. This relative timer phase is then followed by an adder phase of growth that involves synthesis of new cell wall material for making daughter cell poles. Cell wall labelling measurements reinforce this biphasic model of growth, showing that a crossover from uniform lateral growth to localized cell-wall growth regulate the timing of cell division. Banerjee et al further proposed a mathematical model of this "mixer" mechanism of size control that quantitatively explains the biphasic growth dynamics. The model predicted that the crossover onset from timer to adder is regulated by the growth rate of the bacteria, which can be tuned by environmental variations. This prediction was indeed confirmed by cell growth experiments at other temperature.
Although the molecular basis for biphasic cell size control is unknown, this study provides new insights for investigating the molecular candidates regulating cell size and division control in bacteria. The timer phase may be related to the duration of chromosome replication, which is independent of cell size. On the other hand, the added cell size is related to the volume of the daughter cell poles that is independent of the mother cell volume at birth.
 Close
Close

