Various compounds can reduce the activity of enzymes. They may act in a variety of different ways, and indeed may be reversible or irreversible inhibitors of the enzyme.
On this page there are notes about:
An irreversible inhibitor causes covalent modification of the enzyme, so that its activity is permanently reduced. Compounds that act as irreversible inhibitors are often useful as drugs that need be taken only every few days, although adjusting the dose to suit the patient’s response is a lengthy process with such compounds. By contrast, the effect of a reversible inhibitor can be reversed by removing the inhibitor, e.g. by dialysis or gel filtration.
The normal sequence of an enzyme reaction can be represented as:
![]()
where:
E = enzyme
S = substrate
E-S = enzyme-substrate complex
E-P = enzyme-product complex
P = product
There are three main types of reversible inhibitor:
They interact with the enzyme or enzyme-substrate complex at different stages in the sequence
A competitive inhibitor competes with the substrate for the active site of the enzyme:
![]()
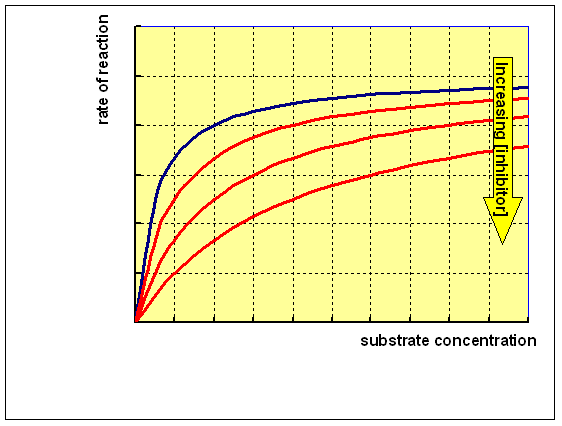
This means that increasing the concentration of substrate will decrease the chance of inhibitor binding to the enzyme. Hence, if the substrate concentration is high enough the enzyme will reach the same Vmax as without the inhibitor. However, it will require a higher concentration of substrate to achieve this and so the Km of the enzyme will also be higher. Reacting the enzyme with a range of concentrations of substrate at different concentrations of a competitive inhibitor will give a family of curves as shown below:
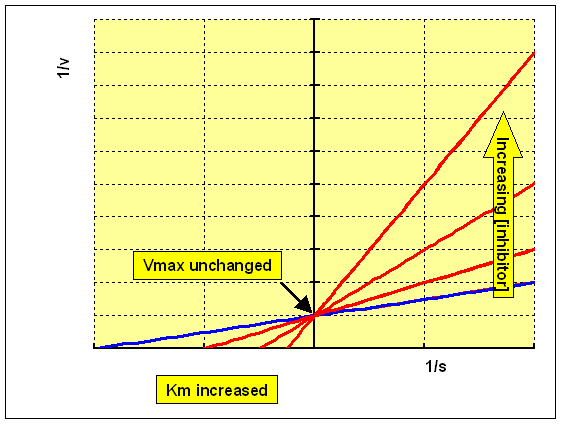
The Lineweaver-Burk double reciprocal plot for this set of data shows a series of lines crossing the y (1/v) axis at the same point - i.e. Vmax is unchanged, but with a decreasing value of 1/Km (and hence a higher Km) in the presence of the inhibitor:
A non-competitive inhibitor reacts with the enzyme-substrate complex, and slows the rate of reaction to form the enzyme-product complex.
![]()
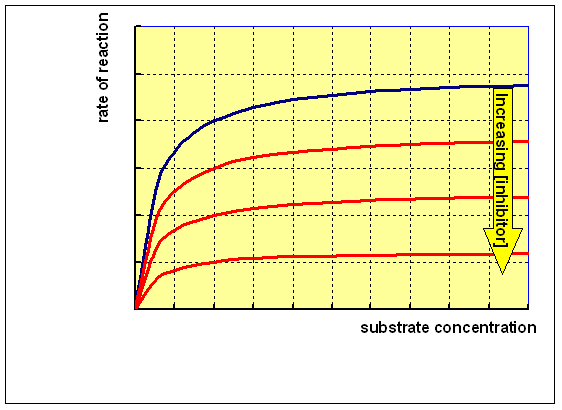
This means that increasing the concentration of substrate will not relieve the inhibition, since the inhibitor reacts with the enzyme-substrate complex. Reacting the enzyme with a range of concentrations of substrate at different concentrations of a non-competitive inhibitor will give a family of curves as shown below:
The Lineweaver-Burk double reciprocal plot for this set of data shows a series of lines converging on the same point on the X (1/S) axis - i,.e. Km is unchanged, but Vmax is reduced:
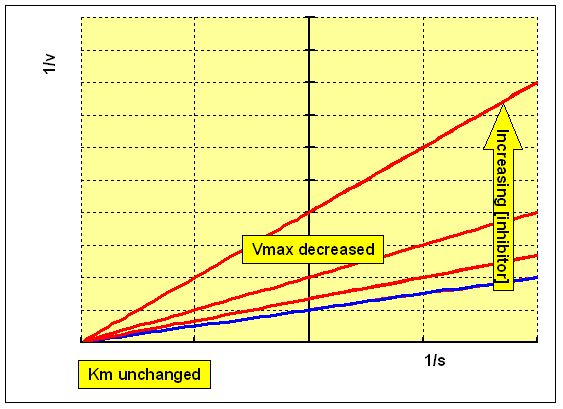
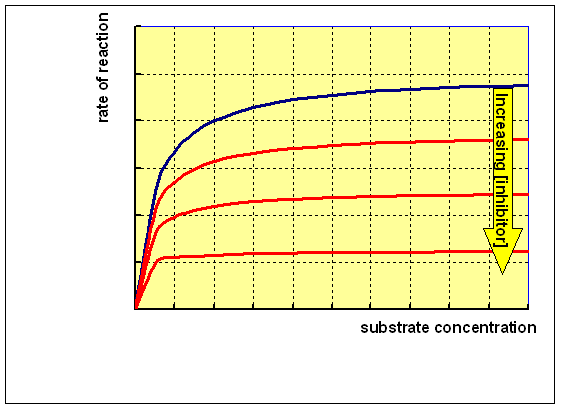
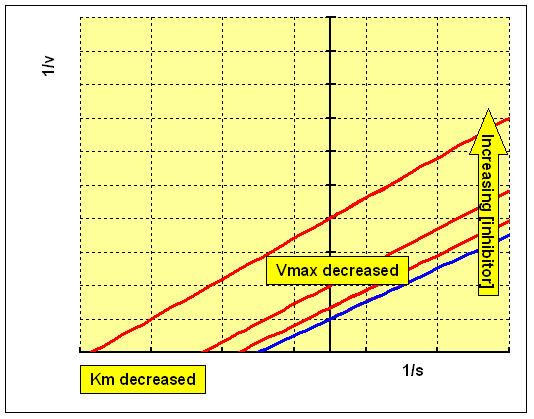
This is a very rare class of inhibition. An uncompetitive inhibitor binds to the enzyme and enhances the binding of substrate (so reducing Km), but the resultant enzyme-inhibitor-substrate complex only undergoes reaction to form the product slowly, so that Vmax is also reduced:
![]()
Reacting the enzyme with a range of concentrations of substrate at different concentrations of an uncompetitive inhibitor will give a family of curves as shown below:
The Lineweaver-Burk double reciprocal plot for this set of data shows a series of parallel lines - both Km and Vmax are reduced:
If the requirement is to increase the intracellular concentration of the substrate, then either a competitive or non-competitive inhibitor will serve, since both will inhibit the utilisation of substrate, so that it accumulates.
However, if the requirement is to decrease the intracellular concentration of the product, then the inhibitor must be non-competitive. As unused substrate accumulates, so it will compete with a competitive inhibitor, and the final result will be a more or less normal rate of formation of product, but with a larger pool of substrate. Increasing the concentration of substrate does not affect a non-competitive inhibitor.
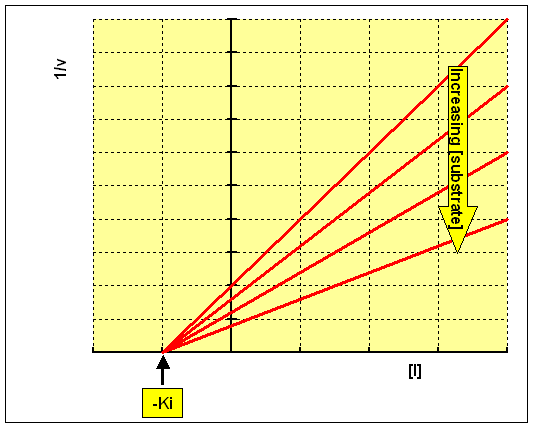
The inhibitor constant, Ki, is an indication of how potent an inhibitor is; it is the concentration required to produce half maximum inhibition.
Plotting 1/v against concentration of inhibitor at each concentration of substrate (the Dixon plot) gives a family of intersecting lines.
For a competitive inhibitor, the lines converge above the x axis, and the value of [I] where they intersect is -Ki
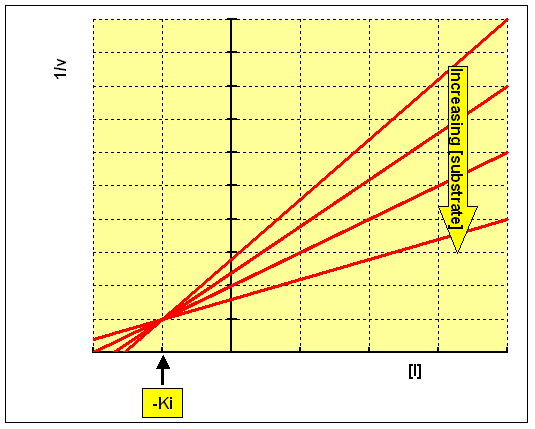
For a non-competitive inhibitor, the lines converge on x axis, and the value
of [I] where they intersect is -Ki