Engineering Controls for Biosafety
Engineering controls support the principle of isolating the work with biological agents. They include specific equipment, infrastructure and design elements.
This page details the types of engineering controls used at UCL.
UCL Estates work closely with departments that work, or plan to work, in containment labs to ensure that all required engineering controls are in place. Administrative controls should be inplace for to ensure maintainance and inspections are carried out on engineering controls to protect people the environment as well as meet legal compliance for statutory testing. For specific information on CL3 areas contact the local CL3 or Safety Services.
Surfaces
Surfaces should be impervious to water, resistant to acids, alkalis, solvents, disinfectants and decontamination agents and easy to clean. This is not only in the design stage but should be included in the maintenance of the area and applies to all areas that work with any biological agent. For example:
- wood on doors or window frames must be treated to keep it impervious to both liquids and gases, through annual painting or treatment.
- floors must be checked to ensure rips, cuts and other imperfections are managed.
Microbiological Safety cabinets (MSCs), isolators and other containment for aerosols
Microbiological Safety Cabinets (MSCs) and isolators are both forms of Local Exhaust Ventilation providing containment of biological agents and protection to the user when used correctly. MSCs should be used for any work potentially producing an infectious aerosol. Users should be trained in how to safely use an MSC and understand what to do if there is a fault alarm or emergency.
A risk assessment should be in place to manage containment of activities that cannot be carried out in an MSC e.g. Centrifugation.
Types of Containment device
Class 1 MSC
Class 1 These can be used for biological agents from Hazard group 1-3 and GMM class 1-3, The class 1 MSC works by pulling unfiltered air from the laboratory environment and the airflow is then pulled through a HEPA filter ducting to be extracted through ducting. Class 1 MSCs are good for protecting the user but do not offer sterility for the material being used inside. The picture below shows the flow of air with red and blue arrows. The red arrows show the unfiltered air flow as it is pulled into the MSC. The blue arrows show the HEPA filtered airflow to the external environment. The aerosolised microbiological contaminants will be captured by the HEPA filtered, thus preventing a release to the environment. This is providing they are maintained and tested regularly.
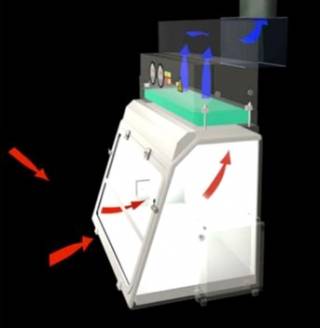
Class 2 MSC
Class 2 MSCs can also be used for HG 1-3 microorganisms and Class 1-3 GMOs. A Class 2 MSC also pulls air into the MSC but the air from the surrounding laboratory is pulled under the workspace inside the MSC, up the back of the MSC to the plenum where it is HEPA filtered before 70% of the filtered air is returned to the working space inside the MSC through a diffuser creating a clean curtain of air within the workspace of the MSC. The remaining 30% of filtered air is extracted either to the external environment if the MSC is connected to the building HVAC. If not ducted 30% of the filtered air is returned to the lab.
These cabinets offer protection to the user and to the materials in the MSC provided they are used correctly. The airflows are sensitive to sudden movements which should be avoided. There are limitations on chemical usage because they do not protect the user from chemicals. Solvents should be limited to avoid the build-up of a flammable atmosphere. A non-ducted class 2 MSC is not suitable for chemicals with strong and toxic or harmful vapours / mists. Limited amounts of these chemicals can be used in a ducted Class 2. Local procedures will need to be in place for the fumigation of MSCs not connected to the building ducting as the removal of fumigant will need to be done safely.
The picture below shows the flow of air with red and blue arrows. The red arrows show the unfiltered air flow as it is pulled into the MSC. The blue arrows show the HEPA filtered airflow where 70% of this filtered air is sent to the work space via a diffuser, proving a clean curtain of air protecting the samples and the user, whilst the remaining 30% is either returned to the laboratory or sent external to the building if connected to the building duct work.
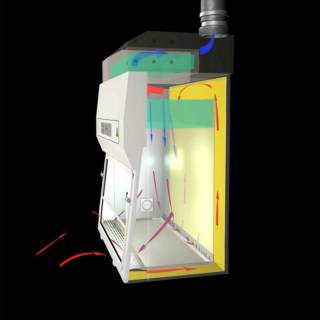
Class 3 MSC / Isolators
Class 3 MScs / Isolators are enclosed with HEPA filters on the inlet and extract air. The operator wears gloves / gauntlets that are attached to sealed ports integral to the cabinet which avoids exposure to the hazardous materials being used. These are often used in CL4 facilities to protect people and the environment from the highest hazard biological agents.
The picture below shows the flow of air with red and blue arrows. The red arrows show the unfiltered air flow as it is pulled into the Class III MSC isolator. The blue arrows show the HEPA filtered airflow entering and exiting the Class 3 isolator.
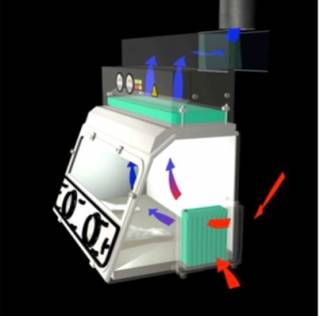
Siting of MSCs
British Standard 5726:2005 details the siting of MSCs. In summary, they should be sited away from heat sources, vibration sources e.g. centrifuges, drafts from doors, windows and people walking past, fume cupboards and equipment that could vibrate such as centrifuges to avoid disruption of the protective air flows especially with a Class 2 MSC. MSCs should be sited so that fire detection or lighting are not impeded.
Once in situ an MSC should be tested to ensure it is providing protective airflows, KI disc testing provides data on protection factors and DOP testing will test the integrity of the HEPA filters in the MSCs. Local testing using anemometers can be used to confirm the airflows in an MSC. There should be local procedures on how to carry this out.
Autoclaves
Best practice requires consideration of the location of autoclaves so that isolation of the hazard can be carried out. The baseline standard is for autoclave to be;
- available for Class 1 Genetically Modified Organism (GMO) and Hazard Group (HG) 1 waste
- within the building for Class 2 GMO and HG2 waste.
- Within the CL3 suite for Class 3 GMO and HG3 waste
Each autoclave is different and information should be available on what cycle to run and why, including information on how to stack items in the machines to achieve the best effect.
All autoclaves should be asset registered to ensure they recieve a thorough examination and validated for the waste they are expected to inactivate.
Negative pressure and airlock
Negative pressure is a requirement for CL3 labs to contain potentially infectious aerosols. The pressure in a CL3 laboratory should be negative to the immediate surroundings. Entry is typically via an airlock which allows for a cascade of negative pressure. Other labs may require this control depending on the work carried out and the precautions required. The design of any lab that will require containment must include whether this control is necessary and how it will be achieved.
In most cases, the failure of the negative pressure is a safety-critical failure and arrangements should be in place to minimise the likelihood and impact of any potential failure.
HEPA filters
HEPA (high-efficiency particle air) filters theoretically remove at least 99.97% of dust, pollen, mould, bacteria, and any airborne particles with a size of 0.3 microns (µm). HEPA filters are placed into Microbiological Safety Cabinets to remove microorganisms being released back into the environment.
In a CL3 laboratory the extract air should be HEPA filtered to avoid contaminating the environment with HG 3 microorganisms or Class 3 GMOs. At Containment level 4 HEPA filters are required in the intake and extract air. This reduced the risk of release of high hazard biological agents being released into the environment.
The same filtration on incoming and outgoing air, helps to provide a clean room environment.
Sealing for fumigation
MSCs and CL3 laboratories should be sealable to allow for fumigation. Routine fumigation of CL3 laboratories is not mandatory according to the UK Government's Advisory Committee on Dangerous Pathogens (ACDP) guidance. However, there is a requirement to be able to safely fumigate a facility in the event of a spillage or leakage which could lead to widespread contamination.
Incinerators
Incinerators should be available for the disposal of carcasses. These do not need to present in the building, and risk assessments should cover how the carcass will travel to the incinerator or what other disposal arrangements are in place.
Last updated: Tuesday, January 17, 2023
 Close
Close


