We have several topics of research focus:
Hypoxia
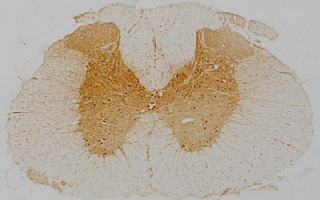
Particular current interests derive from our discovery that neuroinflammatory lesions can be hypoxic, and sufficiently hypoxic to impair mitochondrial metabolism. This impairment is especially likely in an inflammatory environment containing nitric oxide and superoxide. The hypoxia and energy deficit directly contribute to three of the cardinal features of MS, namely loss of function (symptoms), demyelination and degeneration. Based on this realisation, our research explores novel protective treatment strategies that we are developing for clinical trial. One approach is to reduce energy demand by partial blockade of sodium channels. This therapy is also effective in dampening the activation of microglial cells (these cells can damage brain tissue), and we have recently shown the efficacy of one clinically relevant drug in this respect. Therapies based on this new understanding have been examined in recent and ongoing clinical trials in MS and related diseases. Encouragingly, such therapies can be very effective in neuroprotection, and they are also inexpensive and safe for long term administration.
Image: Inflamed spinal cord showing strong labelling for hypoxia in the grey matter.
Blood Flow
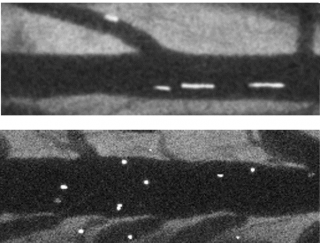
Image: Blood flow in vessels indicated by fluorescent streaks. Long streaks (upper image) show fast flow through normal tissue, and short streaks (lower) show slow flow through inflamed tissue.
Mitochondria
Mitochondria are of particular interest in our research. In the normal nervous system we have found that mitochondrial trafficking along axons is highly influenced by the level of impulse activity along the axons, which has implications for neurodegenerative disease. In the inflamed nervous system we have discovered that many axonal mitochondria become non-functional, which starves the axons of energy. In fact our evidence is that the energy insufficiency can cause conduction failure (and hence symptoms) and ultimately degeneration. We have also investigated how mitochondria become damaged in diabetic axons, and how this damage contributes to diabetic neuropathy.

Image: Retinal vasculature revealed as a shadow over green fluorescence due to ongoing retinal mitochondrial respiration.
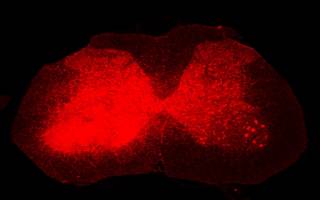
Image: Excessive superoxide production in inflamed tissue on the left of the spinal cord.
Model of the early MS lesion
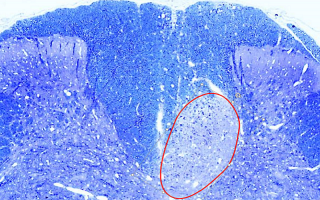
Image: Pattern III demyelination (circled), as occurs in early MS lesions.
Model of ‘slow-burning’ neurodegeneration
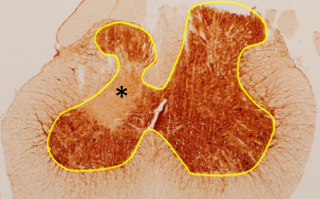
Image: Grey matter atrophy (left) and mitochondrial failure (asterisk) in a lesion of slowly progressive neurodegeneration.
Techniques employed
- Confocal microscopy to monitor mitochondrial dynamics and membrane potential (a measure of mitochondrial metabolism/health), blood flow, tissue metabolism and inflammation in real time
- Electrophysiology to monitor changes in neurological function
- Light and electron microscopy, including a range of immunohistological methods to determine the metabolic and cellular consequences of inflammation
- Tissue oxygen monitoring by ratiometric oxygen sensitive fluorescent tracer, optical probe and immunohistochemical methods
- Near-Infrared Spectroscopy (NIRS) using non-invasive methods to monitor mitochondrial function and tissue oxygenation within the brain
- Multispectral Imaging for non-invasive monitoring of retinal tissue oxygen concentration
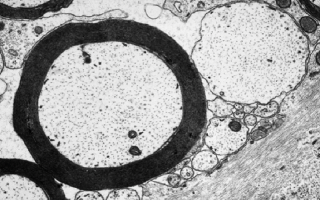
Image: Electron microscope image showing a normal myelinated axon (left) and a demyelinated axon (right).
 Close
Close

