Rohan de Silva is a Reader in Neuroscience.
Rohan was at University in Bern, Switzerland where he completed a Licentiate (MSc) in Biochemistry and Molecular Biology. This was followed by a DPhil in Biochemistry at the University of Oxford under Professor Gordon Lowe, FRS. With his first postdoctoral fellowship he did his first work on the genetics of Alzheimer’s disease at Duke University Medical Center under the late Professor Allen Roses. After brief detours into bladder cancer and cytokines in Alzheimer’s disease, Rohan joined the Reta Lila Weston Institute under Professor Andrew Lees continued molecular genetics studies of neurodegeneration with focus on the tau gene and protein. Rohan and group have made contributions into the understanding of the functional basis of the contribution of tau to the risk of neurodegeneration, particularly in progressive supranuclear palsy and corticobasal degeneration.
Our research
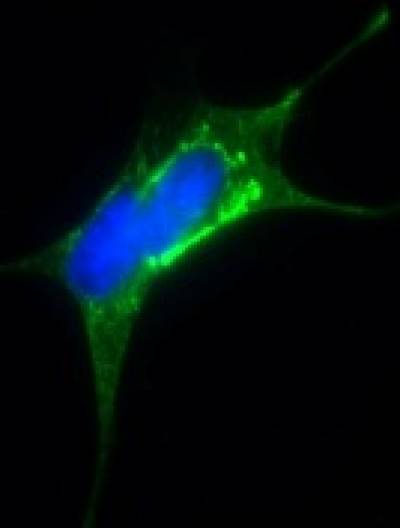
Tau has emerged as an important driver of the large class of neurodegenerative disorders that are collectively called the “tauopathies”. They are defined by microscopic inclusions consisting of clumps (tangles) of the tau protein within the nerve cells (see picture). It is recognised that the process of conversion of normal tau protein, important for normal nerve cell function, into the tangle, could be a crucial driver of progression of the tauopathies. It is therefore plausible that future therapies that target tau would be effective in more than one of these devastating disorders.
Tau protein is essential for the structural integrity and function of nerve cells. Defects in the tau due to tau gene mutations directly lead to neurodegeneration in the frontotemporal dementias. Excessive cellular levels of normal tau due to subtle genetic risk variation and disturbances in cellular milieu lead to the meeting of the unemployed tau molecules and clumping to form the tangles.
The primary focus of our group has been the investigation of the genetic and cellular factors that regulate and affect production (expression) of the tau protein. Specifically, these include:
- Understanding the effect of inherent common genetic variation on regulation of tau gene expression.
- Identifying other genes and molecules that regulate tau gene expression.
- Unravelling the essential molecular events that initiate the process of clumping (aggregation) of tau.
- Developing molecular therapeutic tools to modulate tau gene activity and prevent or slow tau aggregation
Our overriding goal is to identify tau-based therapies that could slow or stop pathological progression in tauopathies.
Group Members:
Postdoctoral fellows:
Dr Roberto Simone: Role of long non-coding RNA in tau gene expression
Drs Ezat Sajedi and Ioanna Sevastou: Tau-bases therapies
PhD Student:
Ms Bimali Hapuarachchi: The role of UPR and PERK in tauopathies.
Technician/PhD student:
Transcript diversity and allele-specific expression of the PSP-associated MOBP gene.
Recent Publications:
Šimić G, Babić Leko M, Wray S, Harrington C, Delalle I, Jovanov-Milošević N, Bažadona D, Buée L, de Silva R, Di Giovanni G, Wischik C, Hof PR.
Biomolecules. 2016 Jan 6;6(1):6. doi: 10.3390/biom6010006. Review.
Vuono R, Winder-Rhodes S, de Silva R, Cisbani G, Drouin-Ouellet J; REGISTRY Investigators of the European Huntington’s Disease Network., Spillantini MG, Cicchetti F, Barker RA.
The role of tau in the pathological process and clinical expression of Huntington's disease.
Brain. 2015 Jul;138(Pt 7):1907-18. doi: 10.1093/brain/awv107.
Magdalinou NK, Paterson RW, Schott JM, Fox NC, Mummery C, Blennow K, Bhatia K, Morris HR, Giunti P, Warner TT, de Silva R, Lees AJ, Zetterberg H.
J Neurol Neurosurg Psychiatry. 2015 Nov;86(11):1240-7. doi: 10.1136/jnnp-2014-309562.
Swirski M, Miners JS, de Silva R, Lashley T, Ling H, Holton J, Revesz T, Love S.
Alzheimers Res Ther. 2014 Dec 1;6(5-8):77. doi: 10.1186/s13195-014-0077-y.
Ferrari R, Ryten M, Simone R, Trabzuni D, Nicolaou N, Hondhamuni G, Ramasamy A, Vandrovcova J; UK Brain Expression Consortium., Weale ME, Lees AJ, Momeni P, Hardy J, de Silva R.
Neurobiol Aging. 2014 Jun;35(6):1514.e1-12. doi: 10.1016/j.neurobiolaging.2014.01.010.
 Close
Close