Find out about Dr Matthew Reeves' research on viral and cellular factors controlling HMCV latency and reactivation.
Our studies seek to understand the cellular and molecular basis of HCMV latency and reactivation. A comprehensive understanding of the viral and cellular factors contributing to HCMV latency and reactivation will underpin new therapeutic strategies to tip the balance in the hosts’ favour to reduce pathogenesis and associated disease.
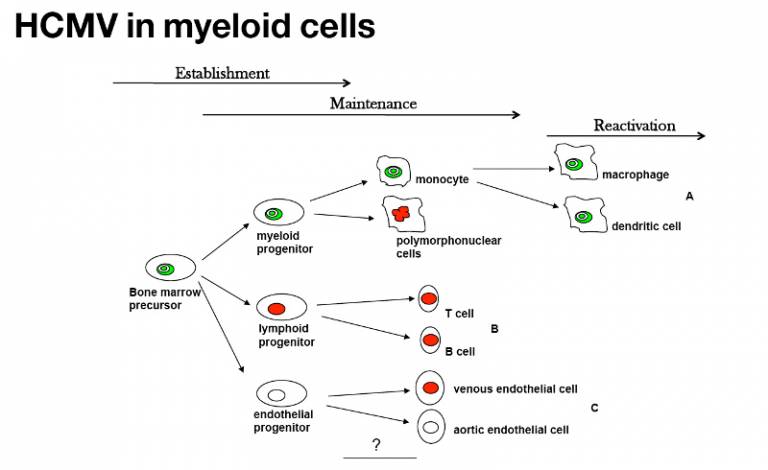
HCMV infects CD34 bone marrow progenitor cells which are the precursor of multiple cell types. Despite this, the latent viral genome is restricted to the monocyte/myeloid lineage. The mechanisms that govern this restriction to the myeloid lineage are unclear. They could involve viral functions supporting myelopoiesis, or it could be that myeloid cells are an inherently favourable environment for HCMV latency.
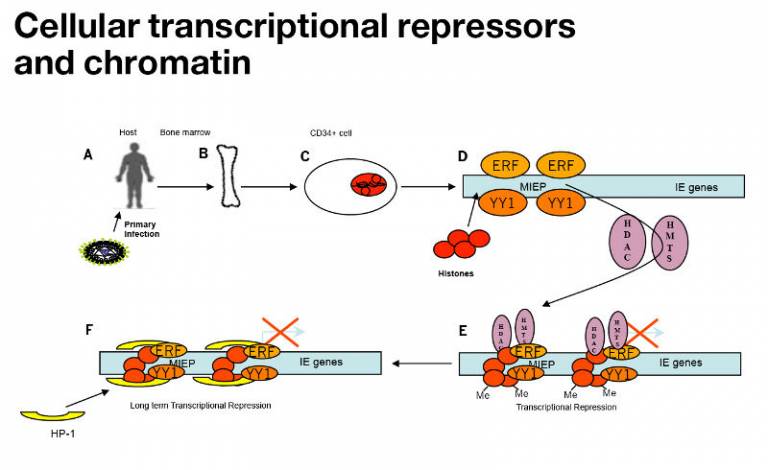
The molecular basis of HCMV latency is argued to involve cellular elements of transcriptional regulation. Repression of the major immediate early promoter (MIEP) is key for establishing latency and the action of higher order chromatin structure is central to this and is a common theme in herpes biology.
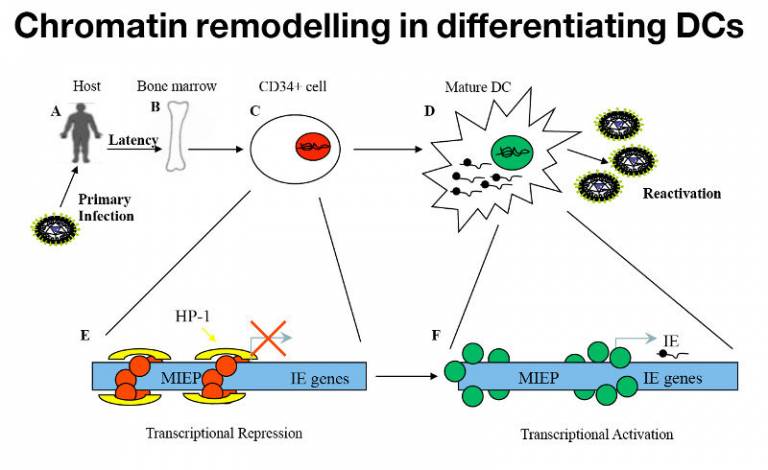
Cellular differentiation of myeloid progenitors promotes reactivation in DCs. The reactivation phenotype is consistent with chromatin remodelling. Understanding how chromatin provides the platform to interpret celllar signalling and trigger reactivation is thus central to our studies.
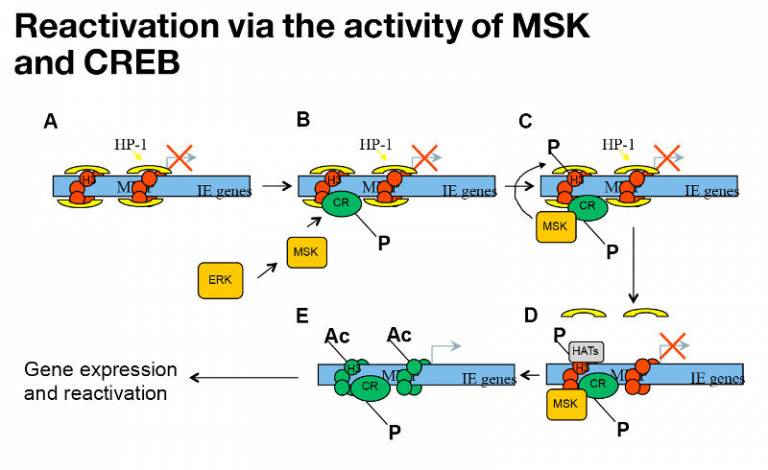
Our current working model is that cellular kinases are recruited to the MIEP in a CREB dependent manner. This drives the chromatin remodelling required to trigger IE gene expression. Why this occurs in DCs specifically is an area of study.
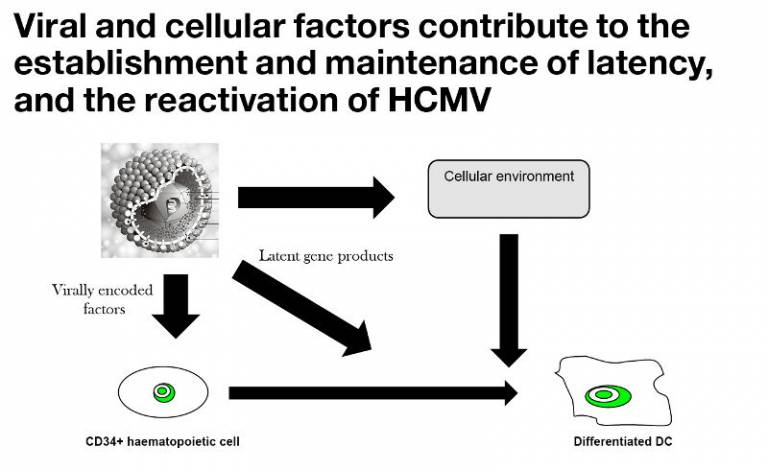
Ultimately our interests lie in understanding the viral manipulation of the cellular environment to establish latency and reactivate. These processes involve both cellular and viral functions that are active throughout latency or become activated at key points through the latent/reactivation viral lifecycle.
 Close
Close

