Helicosphaera carteri cell section and coccosphere
- Coccolithophores
-
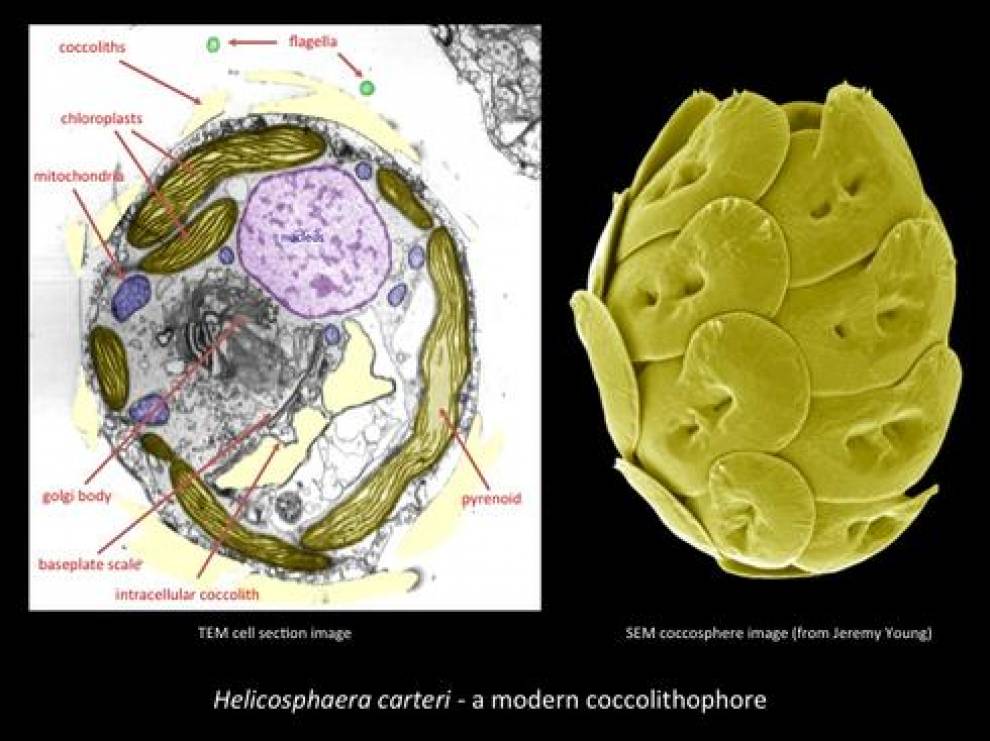
The living coccolithophores are marine, unicellular, flagellate phytoplankton, belonging to the phylum Haptophyta and division Prymnesiophyceae. Haptophyte algae have golden-brown chloroplasts, an exoskeleton of scales and a unique flagella-like structure, known as the haptonema. Within the Haptophyta, only the coccolithophores produce calcified scales know as coccoliths. Coccolithophores produce two types of disc-like coccoliths, heterococcoliths and holococcoliths, which are distinguished by their morphology, mode of formation, and life-cycle stage during which they are produced. They can also produce non-coccolith structures, such as ceratoliths (e.g. Ceratolithus) and pentaliths (e.g. Braarudosphaera), which diverge from the typical disc-like form. Many extinct non-coccolith (nannolith) fossils may also have been produced by coccolithophores.
- Coccoliths
-
Heterococcoliths are typically circular to elliptical discs or rings constructed from one or more radial arrays (cycles) of elaborate and variably shaped crystal units. This disc or ring is termed the rim and encloses a central area, which may be open, virtually closed or spanned by a variety of structures. Heterococcoliths are formed within the cell (in Golgi vesicles) and then extruded to form a composite exoskeleton, the coccosphere.
Holococcoliths are typically disc- or dome-shaped and formed of numerous, minute (~0.1µm), equidimensional calcite crystallites of simple, usually rhombic, shape. Compared to heterococcoliths, holococcolith morphologies are relatively similar through time, and there is a less well-defined distinction between rim and central-area structures. Holococcolith formation is not well understood but they tend to form in the motile, life-cycle stage and calcification appears to be extracellular. The fossil record of holococcoliths is sporadic and sparse, because their structure is weaker than the heterococcoliths and the small, modern forms rarely survive in sea floor sediments.
- Coccolithophores Life Cycles
-
Our knowledge of haptophytes life cycles is limited to the few species that survive in laboratories, and even these species rarely give up their secrets easily. Like many algae, haptophytes can reproduce asexually or sexually, and can switch between haploid (half the normal DNA compliment) and diploid (complete DNA compliment) phases. Haptophytes are unusual, however, in that they can maintain reproducing populations in both haploid and diploid states and survive this way for many years in laboratories. They obviously switch from one phase to another in the natural environment but we know little of why and how this happens, although it may allow them to resist viral attack or respond to detrimental environmental changes. In some coccolithophores this switch is also accompanied by production of a completely different coccosphere, most commonly haploid motile phases producing holococcoliths and motile and non-motile diploid phases producing heterococcoliths. They do not always conform to this pattern, however, and additional phases may occur, such as benthic stages in near-shore species, and non-coccolith structures or naked stages may also occur.
- Coccolithophore Ecology
-
Living coccolithophores have a widespread oceanic distribution, living in the photic zone and being most diverse at low latitudes. Generally speaking, they are rare or absent at latitudes higher than 70° and flourish in warm, stratified, oligotrophic, mid-ocean environments. A number of species have very broad ecological tolerances but only one freshwater coccolithophore has been well documented.
Coccolithophores show distinct biogeographic distribution patterns, defining broad, latitudinal belts or zones. These zones are distinguished by variations in population composition, although a limited number of species show restricted distribution, limited, for example, to the low or high latitudes. These distribution patterns reflect both temperature and nutrient distributions and are linked to large-scale features of oceanic circulation, such as, divergence zones, ocean gyres and seasonal mixing. The major limiting nutrients are nitrate and phosphate, but the abundance of other trace elements and vitamins, such as Fe, Zn, Mn and thiamine, are also thought to be significant. Some coccolithophores may supplement their nutrient requirements by heterotrophy or phagotrophy, using the haptonema to capture food particles. Seasonally stable, oligotrophic, tropical and subtropical mid-ocean gyre environments support the highest diversities, but standing crops are low, reflecting low reproduction rates. Eutrophic conditions, due to upwelling or deep seasonal mixing, often support high standing crops dominated by one or a few species, however diatoms are the most effective eutrophic phytoplankton in present oceans and coccolithophores generally bloom following depletion of silica in the surface waters. Continental shelf and near shore environments also tend to be unstable and eutrophic in nature and support distinctive assemblages, including taxa, which do not live in open-ocean environments, e.g.Braarudosphaera bigelowii and Pleurochrysis carterae.
Coccolithophore communities also show vertical stratification, with distinctive deep photic-zone assemblages occurring in or below the thermocline/nutricline, exploiting a less-competitive low-light, low-temperature, high-nutrient niche, in particular, Florisphaera profunda and Gladiolithus flabellata.
Evidence from the fossil record suggests that coccolithophores have always displayed broadly similar ecological tolerances, and certainly palaeobiogeographic distributions are generally comparable with those of the present.
- Coccolithophore Evolutionary History
-
Calcareous nannofossils first appeared in the Late Triassic (~220 Ma) as abundant but low-diversity assemblages apparently restricted to low latitudes. All but one species of coccolith disappeared during an extinction event at the Triassic/Jurassic boundary (~200 Ma). Nannofossils were rapidly re-established in the earliest Jurassic (185-195 Ma) and appear to have colonized all marine environments during this time. Most major coccolith families were established during the Early Jurassic radiation. Diversity increased steadily through the Jurassic and Cretaceous, reaching a diversity-peak in the Late Cretaceous (~75 Ma). Nannofossils were severely affected by the Cretaceous/Tertiary boundary mass-extinction event (65.5 Ma) when >90% of species became extinct, including virtually all the common species. Subsequently, they recovered rapidly in the Early Paleocene, reaching a second diversity peak in the Middle Eocene (~54 Ma), comparable to that of the Late Cretaceous. The Paleocene radiation was more rapid and diverse than the Mesozoic radiations, and established significantly different coccolith families. There followed a significant decline into the Oligocene, increased diversity in the Miocene and a general decrease into the Pleistocene.
- Nannoplankton Taxonomy
-
Taxonomy is the description, naming and classification of organisms, and the taxonomy of coccolithophores is based largely upon the structure of the coccoliths. The production of coccoliths requires complex biochemical processes, which have been programmed into the group since their origination, and so they provide an apparently faithful record of evolutionary history. The coccolith-based taxonomy has stood up well to the introduction of detailed biological and DNA-based molecular genetic studies on living taxa and in most case the different approaches agree well.
All heterococcolith formation observed to date, begins with the formation of a ring of calcite crystals (the proto-coccolith ring) on an organic base-plate scale within the cell. The proto-coccolith ring is formed of calcite crystals with alternating sub-vertical and sub-radial c-axis orientations, known as the V- and R-units. Their subsequent growth into complex coccolith structures forms the basis for coccolithophore taxonomy.
An online taxonomic resource, Nannotax, can be found at:
An overview of current nannofossil taxonomy is online at:
http://ina.tmsoc.org/taxcatalog/INTRO.HTM
A guide to descriptive terminology is online at:
 Close
Close

