Group Leader: Professor Bart Vanhaesebroeck, PhD FMedSci
We study PI3K enzymes which control signal transduction in cells. PI3K overactivation occurs in cancer, some immune contexts and in overgrowth syndromes.
We aim to understand the mechanism of PI3K action in cells and organisms. Over the years, our studies have contributed to PI3K drug development, clinical trials and drug approval.
As detailed below, a highlight of our work is the discovery of PI3Kdelta. PI3Kdelta inhibitors are now approved for the treatment of leukemia and are also being tested in cancer immunotherapy.
After a decade-long focus of inhibiting PI3K enzymes, we have recently started to develop compounds to activate PI3K, the first paper on which was published recently (rdcu.be/dcUpi). In preclinical studies, these compounds can protect cardiac and neural tissue from the sort of damage seen in heart attack and stroke, and also stimulate nerve regeneration after injury. We aim expand these PI3K activator studies in the near future.
Publications: View full list of publications on PubMed.
Talks: For a general introduction to PI 3-kinase and lipid messengers: https://hstalks.com/bs/461/
Videos of the lab & staff:
- Life as PhD student in the lab - http://youtu.be/iXeUxp_h3Bw
- 30 years in PI 3-kinase research - https://www.medchemexpress.com/webinar-Bart.html
- Our EU-funded PhD Training Network - https://www.youtube.com/watch?v=02Kdk9D5Y3g
- On our successful collaboration with AstraZeneca Open Innovation - https://openinnovation.astrazeneca.com/success_stories.html
Get in touch: We welcome informal enquiries - if you would like to work with us, please contact Bart Vanhaesebroeck bart.vanh@ucl.ac.uk

Our laboratory covers 3 main lines of research:
1. PI3K isoform function: we aim to uncover the functions of the PI3K family members in normal physiology and in disease, and to understand their molecular mechanism of action. The main sponsors of this work are Cancer Research UK, the EU and the BBSRC.
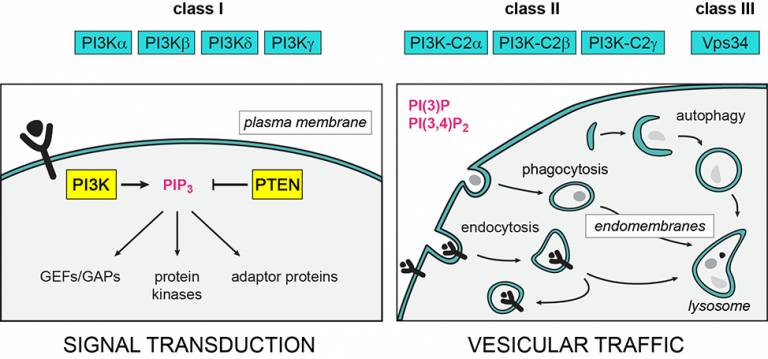
2. PTEN Hamartoma Tumour Syndrome (PHTS): this is a human condition in which the PTEN gene is faulty, predisposing to overgrowth, autism-like spectrum and cancer. PTEN is an enzyme which keeps PI3K in check. We aim to understand how PTEN works in an organism, and to explore if PI3K inhibitors can be used to alleviate PHTS. The main sponsor of these studies is the PTEN Research Foundation.
3. Exploring alternative ways to interfere with PI3K signalling: this is a drug development programme, under the umbrella of the BRC Therapeutic Innovation Network (TIN). Bart V. is the co-Chair of the Small Molecule Therapeutic Innovation Network (TIN), launched in July 2018. The main sponsors of this work are UCL and the NIHR University College London Hospitals Biomedical Research Centre.
A highlight of our work to date is the discovery of PI3Kdelta, a PI3K family member that is highly expressed in white blood cells, as a drug target in various diseases. Our team has been involved in the characterization of PI3Kdelta ‘all the way’, from gene cloning through to the generation of the first mouse models and the development of PI3Kdelta inhibitors (by PIramed UK, acquired by Roche in 2008). Over the years, we have uncovered PI3Kdelta as a drug target in immunity, inflammation and haematological cancers and, most recently, as a target for cancer immunotherapy.
In 2014, a PI3Kdelta inhibitor (idelalisib from Gilead Sciences) was the first PI3K inhibitor to be approved, namely for the treatment of specific B-cell leukaemias. PI3Kdelta inhibitors also show great promise in the 'Activated PI3Kdelta Syndrome' (APDS), a disease due to germline activating mutations in the PI3Kdelta gene. Based on our finding that PI3Kdelta inhibition leads to the activation of the immune system against cancer (Nature 2016:535:580), PI3Kdelta inhibitors are now also being tested in cancer immunotherapy trials in solid tumours (see for example: Nature 2022:605:741 and ClinicalTrials.gov and iOnctura pipeline).
 Close
Close


