Professor Henning Walczak, PhD
Chair: Cell Death, Cancer and Inflammation Research Group
Research focus
Research in the Walczak Laboratory is focused on cell death and ubiquitin in inflammation, cancer and immunity. The lab is particularly interested in unravelling the mechanisms on how different death receptor-ligand systems such as the TNF, Fas (CD95) and TRAIL systems are regulated and how they impact cancer cell survival, cancer-related inflammation and immunity. The research aims are to develop novel cancer therapies by specifically inducing cancer cell death and by therapeutically directing the type of death induced in cancer cells to convert cancer-related inflammation from being immune-regulatory to enabling the immune system to recognise and kill cancer cells.
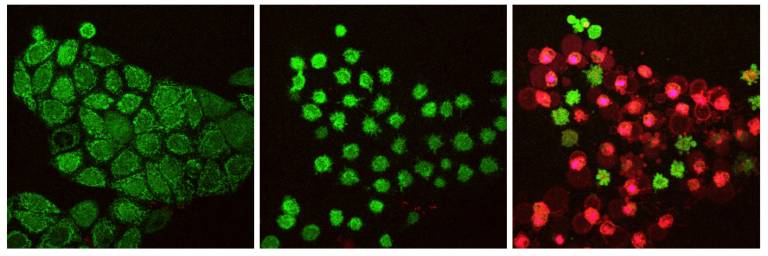
Fig.1 Human cancer cells undergoing TRAIL-induced cell death.
Selected publications
1. Annibaldi A, Walczak H. Death Receptors and Their Ligands in Inflammatory Disease and Cancer. Cold Spring Harb Perspect Biol. 2020 Jan 27. pii: a036384. [Epub ahead of print] PMID:31988141
2. Peltzer N, Walczak H. Cell Death and Inflammation - A Vital but Dangerous Liaison. Trends Immunol. 2019 May; 40(5):387-402. Epub 2019 Apr 16. Review. PMID: 31003931
3. Lafont E, et al, Walczak H.- TBK1 and IKKε prevent TNF-induced cell death by RIPK1 phosphorylation. Nat Cell Biol. 2018 Dec;20(12):1389-1399.
4. Taraborrelli L, et all, Walczak H. LUBAC prevents lethal dermatitis by inhibiting cell death induced by TNF, TRAIL and CD95L. Nat Commun. 9(1):3910, 2018.
5. Peltzer N, et al, Walczak H. LUBAC is essential for embryogenesis by preventing cell death and enabling haematopoiesis. Nature. 557(7703):112-117, 2018.
6. von Karstedt, S., Montinaro, A., Walczak, H. Exploring the TRAILs less travelled: TRAIL in cancer biology and therapy. Nature Reviews Cancer, 17: 352-366, 2017.
7. von Karstedt, et al, Walczak, H Cancer cell-autonomous TRAIL-R signaling promotes KRAS-driven cancer progression, invasion, and metastasis. Cancer Cell, 27: 561-573, 2015.
Latest news
Video: The Alexander von Humboldt Professorship Awards 2019 - Professor Walczak discusses his research, which focuses on the mechanism of programmed cell death.
Features:
New drug target could control onset of neurodegenerative and inflammatory disease
Study team, led by Henning Walczak, have discovered a new checkpoint that prevents a certain type of cell death from occurring. The findings could help scientists and doctors further understand – and potentially slow down or even halt – the progression of auto-immune and neurodegenerative diseases.
Accelerating Immunotherapy - £5 million award to new research collaboration led by Professor Henning Walczak
Cancer Research UK has awarded one of the first 2015 Network Accelerator Awards to the Cancer Research UK-UCL Centre based at UCL Cancer Institute. The award brings together a network of immunologists, cancer biologists and cancer immunotherapists across Centres and Institutes in London, and will also help the next generation of scientists to develop careers in the field.
Body’s defences hijacked to make cancers more aggressive
UCL scientists have discovered that a vital self-destruct switch in cells is hijacked - making some pancreatic and non small cell lung cancers more aggressive, according to research published in Cancer Cell. The team, from the Cancer Research UK Centre at the UCL Cancer Institute, found that mutations in the KRAS gene interferes with protective self-destruct switches, known as TRAIL receptors, which usually help to kill potentially cancerous cells.
Scientists trigger self-destruct switch in lung cancer cells
Cancer Research UK scientists have found a drug combination that can trigger the self-destruct process in lung cancer cells - paving the way for new treatments, according to research presented at the National Cancer Research Institute (NCRI) Cancer Conference.
When healthy cells are no longer useful they initiate a chain of events culminating in self destruction. But cancer cells swerve away from this suicide path and become immortal. This means that cells grow out of control – causing tumours to form.
The Cancer Research UK team, based at the UCL Cancer Institute, has successfully fixed this fault in lung cancer cells – reprogramming the cells to self-destruct.
 Close
Close


