Research is divided into 7 signature programmes and two cross cutting programmes (Chemistry and Imaging Computing) across KCL and UCL.
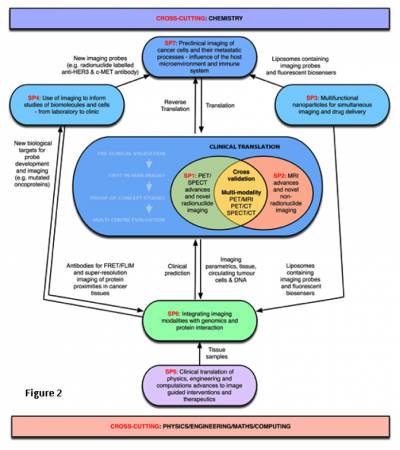
The CCIC is based on seven Signature Programmes:
- SP1: Clinical translation of PET/SPECT advances & novel radionuclide imaging.
This translational programme aims to facilitate personalized medicine by exploiting the synergies of novel, multimodality and multi-parametric imaging methods to improve tumour characterisation, treatment prediction and response assessment in cancer patients. The programme will be underpinned by the use of novel tracer and contrast methodology (SP4) and innovative MRI protocols ( SP2 and SP7).
The unique position of the collaborative KCL/UCL centre in the UK is its technical expertise in PET and MRI development and access to the clinical simultaneous PET-MRI: clinical applications of this new hybrid modality will be explored within this programme.
Leads: Gary Cook and Ashley Groves
- SP2: Clinical translation of MRI advances & novel non-radionuclide imaging.
Developing novel techniques for clinical application is a unique and often unconsidered challenge. Imaging methods that work in the pre-clinical setting need significant time and resource for clinical adaptation. Sequential iterative development, optimisation and modification of techniques is necessary through a series of phantom, normal volunteer, patient volunteer, and feasibility studies before early phase clinical application. Multi-centre evaluation of developing methods requires further optimisation, chiefly of repeatability and reproducibility. This programme provides the clinical translational framework necessary for emerging pre-clinical non-radionuclide imaging methods (SP7) and novel techniques currently available for clinical imaging. This programme will directly feed developments into SP6 (novel imaging parameter predictive modelling for combination with genomics), SP1 (novel MRI sequence developments for simultaneous PET-MRI) and SP5 (image quided therapy).
Lead: Shonit Punwani
- SP3: Multifunctional nanoparticles for simultaneous imaging and drug delivery.
The overall aim of this programme is to develop targeted, self-assembling multifunctional nanoparticles that can simultaneously deliver small molecule therapeutics to primary tumors and metastases, and also deliver optical probes to measure the effects of these therapies at the molecular level. These nanoparticles are designed with appropriate labels for tracking by two or more imaging modalities (PET, SPECT, optical, MR) in vivo. They will be powerful new diagnostic tools that will enable us to answer fundamental questions about the heterogeneity of tumour responses to different treatments. We also aim to take our key EGFR-targeted, nanoparticle-based activity sensing technology, developed in CCIC-I, through to Phase I clinical trials.
Lead: Alethea Tabor
- SP4: Use of imaging to inform studies of biomolecules and cells – from laboratory to clinic
We aim to harness antibody technologies for imaging and therapy of cancer, developing and exploiting novel preclinical and clinical imaging technologies in so doing. Activities within SP4 fall into three areas: design and production of new antibodies (“antibody toolbox”); preclinical evaluation of antibodies in vivo for clinical use; and antibodies as tissue diagnostics (FRET/FLIM and single molecule imaging). These projects interact with each other and SPs 3, 6, and 7, and with the Chemistry cross-cutting programme. Future clinical evaluation of antibodies will be facilitated by combining the UCL cGMP protein production facility with the KCL cGMP radiochemistry and regulatory team (see Chemistry cross-cutting programme).
Leads: Kerry Chester, Barbara Pedley
- SP5: Clinical translation of physics, engineering and computational advances to image-guided interventions.
This programme integrates a significant body of work undertaken within and in association with the CCIC. Our ambition is to translate at least one new image guided application per year to first-in-man studies. This is a significant engineering challenge requiring establishment of a Quality Management System (QMS) to enable CE marking and clinical trials. This pipeline will benefit from the 3 Wellcome/DoH HICF Smart Surgery awards recently started at UCL in image guided laparoscopic liver surgery, image guided prostate biopsy and focal therapy, and in image guided neurosurgery. This Signature Programme will also draw on novel methodology arising from the EPSRC Programme “Intelligent Imaging: Form, Function and Motion across Scale”, led by Hawkes at UCL in collaboration with KCL, ICR and ICL. The work builds on the Nifty suite of open source Medical Image Computing Tools described in the Computational Imaging cross-cutting programme and in particular facilitates the clinical translation of technologies to be developed within SP2 and SP7.
Lead: Dave Hawkes
- SP6: Integrating imaging modalities with genomics and protein interaction analysis to understand cancer genome heterogeneity and predict individualised clinical outcome.
The combination of imaging and genomics is a constant theme across the whole Centre but SP6 is dedicated to integrating imaging modalities with genomics and protein interaction analysis to understand cancer genome heterogeneity and predict individualised clinical outcome.
Our established in-house ErbB/HER dimer imaging (Bublil et al, 2010; Coban et al, 2015; Kiuchi et al, 2014; Nuciforo et al, 2015; Tao et al, 2014), applied to paraffin breast cancer tissues, has shown that 10 years after surgery the HER2-HER3 dimer quantity predicts the likelihood of metastatic relapse independently of HER2 expression (Weitsman et al, 2016). In colorectal cancer, we performed a Bayesian latent class analysis, combined with KRas mutation status and advanced molecular imaging of HER2/3 dimer and reported evidence of two novel groups within a negative trial (COIN, Tim Maughan et al., Lancet 2011). One group had superior progression-free survival and gained a benefit from addition of cetuximab to standard chemotherapy.
As an extension of this work, we have observed that significant correlations exist between some CT image parameters (3D volume) and the HER2-3 dimer quantity determined on tissues from the new EPOC neo-adjuvant cetuximab/oxaliplatin-containing chemotherapy trial (Primrose et al, 2014). Further tissue pathological analyses such as Ki-67 staining are being applied to provide a mechanistic explanation for this correlation. This combined Imaging-genomic-protein interaction analysis is being applied to various trials including LOCATE (prostate), PANTHER (colorectal), the Patritumab Head and Neck Phase II trial, the H2020 MR elastography based programme ‘FORCE’ (breast) and the VALIANT (prostate) in the near future.
In lung cancer, we are using imaging to analyse the subclonal tumour evolution during tyrosine kinase inhibitor (TKI)-resistance in EGFR activation-mutant adenocarcinoma of the lung. This project aims to associate multiregion tumour genomic data generated through whole exome sequencing as part of the TRACERx project with radiomics features extracted from patient CT-PET scans. Genomics features include measures of intratumour heterogeneity (ITH), subclonal evolution and specific actionable genomic aberrations such as mutations in the EGFR gene or the MAP kinase pathway. Furthermore, as patients relapse and receive targeted therapy through the associated DARWIN-I and DARWIN-II clinical studies which are on track to open up patient recruitment in the near future, follow-up scans will be analysed to investigate if resistance can be observed and tracked using a radiomics approach. If successful, automated radiomics analysis of tumour CT-PET scans may be used to rapidly and at very low cost assess the degree of ITH, and possibly to identify likely driver mutations from macroscopic image-based features.
References:
Bublil EM,et al.(2010). Kinase-mediated quasi-dimers of EGFR. FASEB J. 24(12):4744-55. doi: 10.1096/fj.10-166199.
Coban O, et al. (2015). Effect of phosphorylation on EGFR dimer stability probed by single-molecule dynamics and FRET/FLIM. Biophys J. 108(5):1013-26. doi: 10.1016/j.bpj.2015.01.005.
Kiuchi T, et al. (2014). The ErbB4 CYT2 variant protects EGFR from ligand-induced degradation to enhance cancer cell motility. Sci Signal. 7(339):ra78. doi: 10.1126/scisignal.2005157.
Nuciforo P, et al. (2015). Quantification of HER family receptors in breast cancer. Breast Cancer Res. 17:53. doi: 10.1186/s13058-015-0561-8.
Tao JJ et al. (2014). Antagonism of EGFR and HER3 enhances the response to inhibitors of the PI3K-Akt pathway in triple-negative breast cancer. Sci Signal. 7(318):ra29. doi: 10.1126/scisignal.2005125.
Weitsman G, et al. (2016). HER2-HER3 dimer quantification by FLIM-FRET predicts breast cancer metastatic relapse independently of HER2 IHC status. Oncotarget: In press.
- SP7: Imaging cancer cells and their metastatic processes – influence of the host microenvironment and the immune system.
We aim to advance biomedical imaging to map the tumour microenvironment and its inherent heterogeneity, with precision and specificity. Measuring changes in the microenvironment of solid tumours is key to detecting and characterising tumour pathophysiology, in particular precursors to metastasis, and for assessing the distribution of therapeutic agents and a physiological response to therapy. SP7 brings together recent methodological developments offering an important new approach to investigating tumour biology.
Specifically in SP7 we are:
- Imaging cancer cell metastasis using Extra-VAscular Convectography (EVAC) MRI that measures interstitial fluid pressure (IFP) and velocity (IFV).
- Predicting barriers and response to therapy using a comprehensive imaging toolkit, including EVAC; arterial spin labelling (ASL) for measuring blood flow; and diffusion-weighted MRI for probing tissue microstructure, from which the delivery of individual drugs can be predicted, prior to administration.
- Development of the human noradrenalin transporter protein (hNET) and mutant hNET to allow for highly specific imaging of transgenic cells
- Imaging infiltrating T lymphocytes, MSCs and macrophages in brain/lung cancers using optical and radionuclide imaging
Underpinning all SPs will be two key cross-cutting disciplines in the proposed Centre:
- Computational imaging: This programme focuses on image computing & standardisation of techniques and methodology for clinical imaging. It supports development of core software tools and hardware elements, coordinated with international efforts (NAMIC, ITK, IGSTK, MITK and NiftyGuide (part of the NifTK initiative between UCL, KCL, Imperial and ICR)) and in close collaboration with industry (Philips, Siemens, GE, Intuitive, Medtronix, Accuray, VisionRT, Elekta). Leads: Dave Hawkes, Sebastian Ourselin
- Chemistry for better probes: This programme promotes innovative design and synthesis of molecular probes and contrast agents to improve quality, cost and GMP availability, realising the potential of a wide range of radionuclides (C-11, F-18, N-13, Ga-68, Tc-99m, Cu-64/62, Zr-89, Ra-223, Bi-213) and nanoparticle technologies. It also directly supports SPs by synthesising required contrast agents using both novel and conventional approaches. With new BRC translational team support the programme commits to taking 10 new tracers to first-in-man by 2017. Lead: Phil Blower
 Close
Close


