CHC22-dependent trafficking of the insulin-sensitive GLUT4 glucose transporter
Humans have two isoforms of the clathrin heavy chain (CHC), known as CHC17 and CHC22. CHC17 is ubiquitously expressed and is involved in a wide range of intracellular trafficking activities, most notably receptor-mediated endocytosis. In contrast, CHC22 is primarily expressed in muscle cells and performs a more specialised function in trafficking the GLUT4 glucose transporter to an insulin-sensitive, intracellular storage compartment. Whilst CHC17 has been extensively studied, much less is known about how CHC22 operates. One of the main research activities of our lab is utilising a combination of structural biology, biochemistry, genomics and cell biology to elucidate the mechanism, function, and regulation of CHC22 and its dependent pathways.
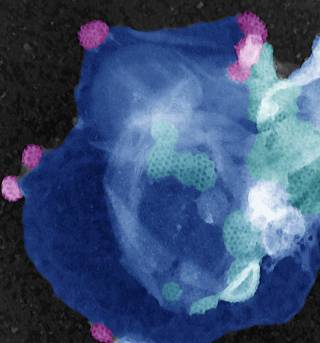
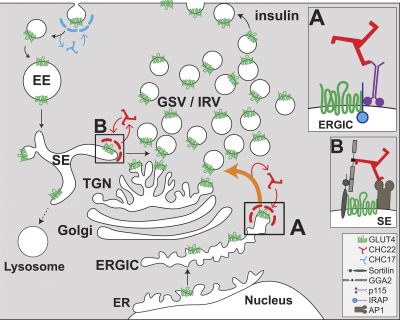
CHC22 clathrin mediates traffic from early secretory compartments for human GLUT4 pathway biogenesis
Camus, Camus, Figueras-Novoa et al. JCB – 2020
- CHC22 traffics newly synthesised GLUT4 from the ERGIC to an intracellular storage compartment.
- This skips the traditional secretory pathway, bypassing the Golgi.
- At the ERGIC, CHC22 forms a complex with p115, IRAP and GLUT4.
- CHC22 forms a complex with GGA2, sortilin and GLUT4 to recycle GLUT4 back to the storage compartment.
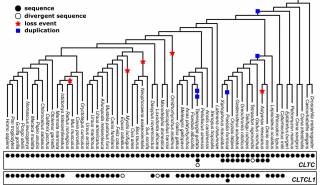
Genetic diversity of CHC22 clathrin impacts its function in glucose metabolism
Fumagalli*, Camus*, Diekmann* et al. eLife – 2019
- CHC22 was independently lost in at least two vertebrate lineages, comprising mice, sheep and pigs.
- Humans possess two high-frequency CHC22 polymorphisms, encoding either a methionine or a valine at residue 1316.
- This polymorphism impacts the dynamics of the CHC22 coat and the ability to sequester GLUT4.
- Changes in the frequency of this genetic variation could be a result of the human transition from a hunter-gatherer to a farming-based diet.
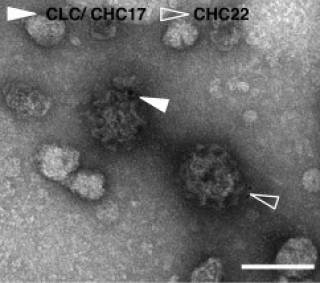
CHC22 and CHC17 clathrins have distinct biochemical properties and display differential regulation and function
Dannhauser*, Camus*, Sakamoto*, et al. JBC – 2017
- CHC22 and CHC17 both form a triskelion and latticed vesicle coats.
- CHC22-coated vesicles are distinct from CHC17 coats.
- CHC22 and CHC17 have different biochemical properties (disassembly and recruitment).
- CHC22 does not support vesicle formation or transferrin endocytosis at the plasma membrane.
 Close
Close